PEG-MGF: Repairing Muscle Tissue Post-Workout
Discover how PEG-MGF repairs muscle tissue after training by activating satellite cells. Complete research guide covering mechanism and benefits for 2026.
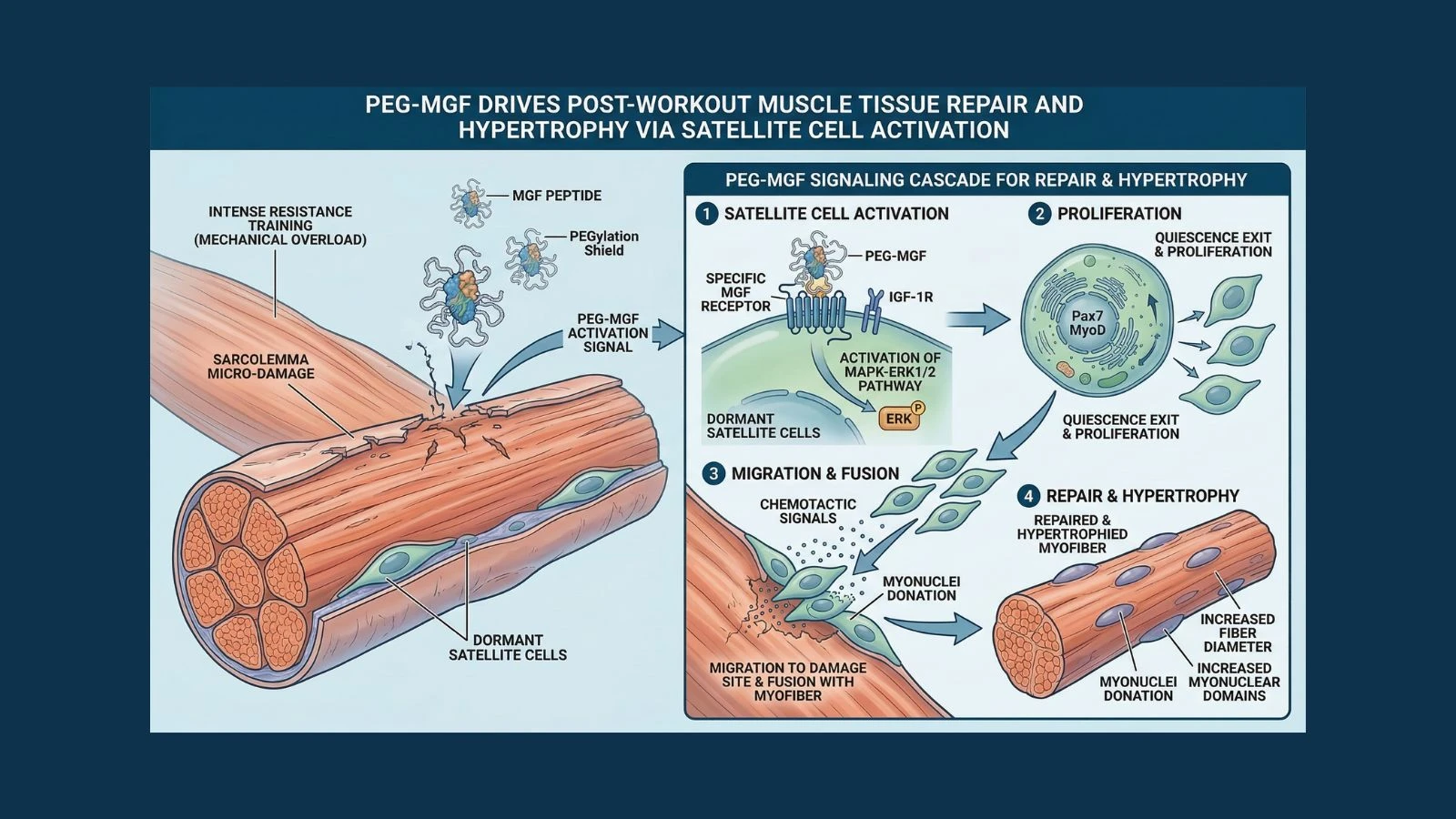
Every hard workout creates damage. That is actually the point.
When you lift heavy weights or push your muscles to their limit, tiny tears form in the muscle fibers. Your body then rushes to repair those tears, and in doing so, it builds the fibers back slightly bigger and stronger than they were before. That process is muscle growth. The damage is not the problem. The speed and quality of the repair is everything.
PEG-MGF is a research peptide designed specifically to accelerate that repair process. It targets the very first step of muscle recovery: waking up the dormant stem cells inside your muscle tissue that do the actual rebuilding work. And it does it in a way that natural muscle growth factors simply cannot match, because PEG-MGF is engineered to stay active in the body long enough to do its job.
For researchers studying muscle hypertrophy, recovery optimization, and age-related muscle loss, PEG-MGF has become one of the most targeted and scientifically interesting tools available. This guide covers exactly how it works, what the research shows, and why the PEG modification transforms a short-lived natural peptide into a sustained and powerful research compound.
New to peptide research? Our Complete Beginner's Guide to Peptide Therapy will give you the foundational knowledge you need before diving into the mechanics of PEG-MGF.
What Is PEG-MGF? Understanding the Science Behind the Name
To understand PEG-MGF, you need to understand its parent molecule first: Mechano Growth Factor, or MGF.
MGF is a splice variant of IGF-1 (insulin-like growth factor 1). When muscle fibers are mechanically stressed, such as during resistance training, a gene called the IGF-1 gene gets activated. Instead of producing standard IGF-1, stressed muscle tissue specifically produces a different version of the protein called MGF. The body uses this local, muscle-specific signal to kick-start the repair process right where the damage occurred.
The problem with natural MGF is that it is extremely short-lived. Once it enters the bloodstream, enzymes break it down within minutes. It barely has time to activate the cells it is supposed to reach before it is gone. This means the repair signal fades fast, and the full potential of MGF's biological activity is never realized.
What Does PEG Stand For?
PEG stands for polyethylene glycol. PEGylation is a well-established pharmaceutical technique where a polyethylene glycol chain is chemically attached to a peptide or protein. This coating does several things at once:
- It shields the peptide from enzymatic breakdown, dramatically extending its half-life in circulation
- It increases the molecular size of the compound, slowing kidney filtration and clearance
- It reduces immunogenicity, meaning the body is less likely to mount an immune response against it
- It allows the peptide to circulate systemically, reaching muscle tissue throughout the body rather than just the local area of injection
The result is a version of MGF that survives long enough to actually do its job. PEG-MGF has a half-life of several days compared to the few minutes of unmodified MGF. This extended activity window is what makes PEG-MGF valuable as a research compound.
PEGylation is the same technology used in several FDA-approved therapeutic proteins. For a broader look at how molecular modifications transform research peptides, our guide on The Future of Peptide Science covers this evolving field in detail.
The MGF Peptide Sequence
The active portion of MGF that drives its biological effects is a 24-amino-acid C-terminal fragment. This Ec peptide region is unique to MGF and is not found in standard IGF-1. It is this specific region that activates satellite cells, the muscle stem cells responsible for tissue repair and growth. PEG-MGF preserves this active fragment while the PEG coating protects it from rapid degradation.
How PEG-MGF Works: The Muscle Repair Pathway
PEG-MGF operates through a specific and well-studied biological pathway. Understanding each step helps explain why it is such a targeted and effective tool for post-workout muscle recovery research.
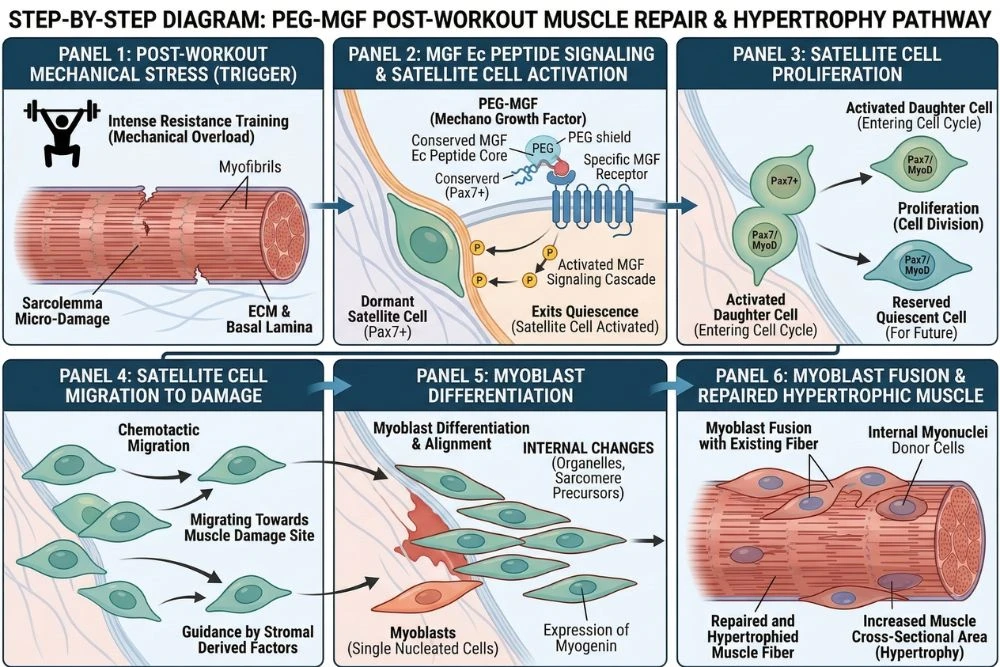
Step 1: Mechanical Damage Triggers the MGF Signal
In a natural context, the MGF pathway begins with mechanical stress on muscle fibers. Resistance training creates micro-tears in muscle tissue. These tears trigger a cellular stress response that activates the IGF-1 gene in muscle cells. Through a process called alternative splicing, the gene produces MGF rather than standard IGF-1 as the immediate local repair signal.
In research settings, PEG-MGF is administered exogenously to mimic and amplify this natural signal, providing a larger and more sustained activation than exercise alone could generate.
Step 2: Satellite Cell Activation
The most critical step in the PEG-MGF pathway is satellite cell activation. Satellite cells are adult muscle stem cells that normally sit in a dormant state attached to the outside of muscle fibers. They are the construction crew waiting for the signal to start work.
MGF's unique Ec peptide region activates these satellite cells directly, causing them to:
- Proliferate: divide and multiply to create a larger repair workforce
- Migrate: move toward the site of damaged muscle fibers
- Differentiate: mature into myoblasts, which are the precursor cells that fuse into muscle fibers
- Fuse: merge with damaged fibers or with each other to form new muscle tissue
Research published in PMC (PubMed Central) confirms that MGF's C-terminal Ec peptide is specifically responsible for satellite cell activation and that this effect is distinct from and complementary to standard IGF-1 signaling, which primarily drives differentiation rather than initial proliferation.
Step 3: MGF vs. IGF-1 Signaling: A Critical Distinction
It is important to understand that PEG-MGF and IGF-1 are not doing the same thing. They work in sequence, not in parallel.
MGF activates and expands the satellite cell population first. It kicks the stem cells out of dormancy and multiplies their numbers. Once the satellite cells have proliferated, IGF-1 then drives their differentiation into mature muscle cells. MGF creates the workforce, IGF-1 trains and deploys them.
This is why combining PEG-MGF with IGF-1 LR3 in research protocols often produces synergistic results. Each compound handles a different phase of the muscle repair and growth process, and together they cover the full repair cascade more completely than either does alone.
For a deep dive into IGF-1's specific role in muscle fiber formation, our guide on IGF-1 LR3 and the Mechanism of Muscle Hyperplasia explains exactly how IGF-1 takes over where MGF leaves off.
Step 4: Systemic vs. Local MGF Activity
Natural MGF acts locally. It is produced at the site of muscle damage and works on the satellite cells nearby. Unmodified MGF does not survive long enough to travel through the bloodstream to other muscles.
PEG-MGF changes this completely. Because its half-life is extended to several days, it can circulate systemically and activate satellite cells in multiple muscle groups, not just the one that was most recently trained. This systemic reach is one of the key advantages of PEG-MGF for research into whole-body muscle repair and recovery.
The Research Benefits of PEG-MGF
The biological specificity of PEG-MGF's mechanism translates into a focused set of research applications. Here is what the published science shows.
Benefit 1: Accelerated Post-Workout Recovery
The most direct application of PEG-MGF research is understanding how to accelerate recovery between training sessions. By supercharging the satellite cell response to exercise-induced muscle damage, PEG-MGF research has shown faster clearance of inflammatory markers, quicker restoration of muscle strength after eccentric damage, and reduced muscle soreness duration in animal models.
The implication for human performance research is significant. Faster and more complete recovery between sessions means more frequent high-quality training is possible without accumulated damage, a concept central to athletic optimization research.
Benefit 2: Muscle Hypertrophy Through Satellite Cell Expansion
Muscle growth over the long term is partly limited by the number of satellite cells available to support fiber repair and expansion. A muscle fiber can only grow so large before it needs additional satellite cell-derived nuclei to support its increased volume. More satellite cells means greater potential for hypertrophic growth.
PEG-MGF's ability to proliferate satellite cells means it directly expands this growth potential. Research in animal models has shown that MGF administration leads to significantly greater satellite cell numbers in treated muscle tissue compared to controls, with corresponding increases in muscle cross-sectional area over time.
Research published in Journals found that local MGF injection in rodent models produced a 25 percent increase in muscle mass over 3 weeks, driven primarily by satellite cell proliferation and fusion, confirming the direct hypertrophic potential of MGF signaling.
Benefit 3: Combating Age-Related Muscle Loss (Sarcopenia)
One of the most compelling research applications for PEG-MGF is the study of sarcopenia, the progressive loss of muscle mass and strength that occurs with aging. Sarcopenia is driven in part by a decline in satellite cell activity and responsiveness to repair signals in older muscle tissue.
Research shows that MGF expression in response to exercise declines significantly with age. Older muscles produce less MGF after training, which contributes to slower and less complete recovery and reduced hypertrophic adaptation. PEG-MGF research explores whether exogenous MGF supplementation can restore the satellite cell response in aging muscle and reverse or slow sarcopenic progression.
A study in PMC Journal of Physiology demonstrated that aged muscle tissue showed significantly blunted MGF expression compared to young tissue after identical mechanical loading, directly linking reduced MGF production to the impaired muscle repair seen in older adults.
Benefit 4: Muscle Preservation During Inactivity
Muscle atrophy during immobilization, such as after injury or surgery, is a major challenge in recovery medicine. Studies have shown that MGF signaling plays a protective role in preventing the rapid muscle loss that occurs when a limb is immobilized.
Animal models using PEG-MGF during periods of induced limb immobilization showed significantly reduced atrophy compared to untreated controls. The satellite cells stimulated by PEG-MGF continued to provide repair and maintenance signals even in the absence of mechanical loading, partially offsetting the atrophic stimulus of inactivity.
Benefit 5: Neurological Protection of Muscle
An emerging and surprising area of MGF research involves neuroprotection. MGF receptors have been found in neural tissue, and research has explored MGF's potential role in protecting motor neurons that innervate muscle tissue. Healthy motor neuron function is essential for muscle activation and coordination, and loss of motor neuron innervation accelerates muscle atrophy in several disease states.
Research from PMC Neuroscience found that MGF administration reduced motor neuron death in models of amyotrophic lateral sclerosis (ALS)-like degeneration, suggesting MGF signaling may have neuroprotective roles that extend beyond direct muscle repair.
PEG-MGF vs. MGF: Why the PEG Modification Matters
Unmodified MGF and PEG-MGF are built from the same biological blueprint, but they behave very differently in research settings. Understanding the differences helps researchers choose the right compound for their specific study design.
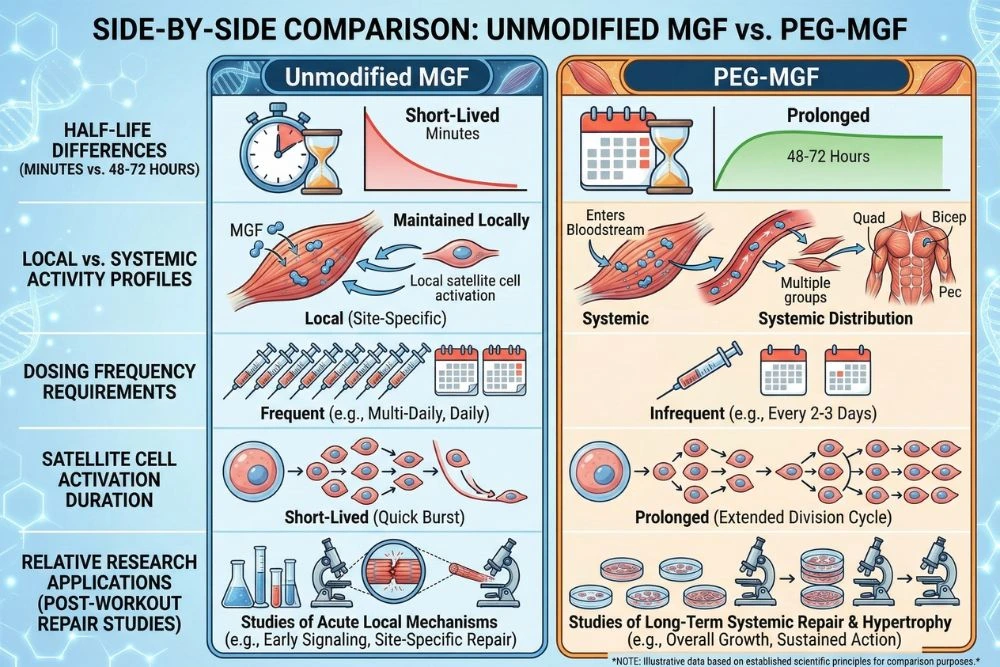
Half-Life: Minutes vs. Days
This is the most fundamental difference. Unmodified MGF has a half-life of just a few minutes in circulation. After subcutaneous injection, it is broken down by circulating peptidases before it can reach distant muscle tissue. It is effectively a local signal only, useful for studying the immediate effects of MGF on tissue directly at the injection site.
PEG-MGF's half-life extends to approximately 48 to 72 hours depending on the PEG chain length used. This allows it to circulate systemically, reach multiple muscle groups, and maintain satellite cell activation over a period of days rather than minutes.
Local vs. Systemic Activity
Unmodified MGF is ideal for localized research, studying the precise effects on a specific muscle group with minimal systemic spread. It is the better choice when researchers want to isolate effects to one tissue location.
PEG-MGF is the better choice when researchers want to study whole-body recovery, systemic satellite cell expansion, or the effects of sustained MGF signaling across multiple muscle groups. Its systemic reach makes it more relevant to practical recovery and hypertrophy research applications.
Dosing Frequency
Because of its short half-life, unmodified MGF typically requires administration immediately post-exercise and sometimes multiple times per day to maintain any meaningful activity window. PEG-MGF only needs to be administered once or twice per week in most research protocols, making it significantly more practical for multi-week study designs.
Research Protocols and Dosing Considerations
Important note: The following information is drawn from published research studies conducted by licensed researchers in controlled settings. PEG-MGF is a research peptide not approved for human use outside of clinical trials. This section is for educational and scientific reference only.
Standard Research Dosing Ranges
PEG-MGF is typically measured in micrograms (mcg) for research purposes. Published studies and well-documented research protocols have used the following ranges:
- Low-range: 100 to 200 mcg per administration, used when studying baseline satellite cell response or in smaller research subjects
- Mid-range: 200 to 400 mcg per administration, the most commonly referenced range in human-oriented research literature
- High-range: Up to 500 mcg per administration, used in research protocols targeting maximum satellite cell expansion or studying dose-response relationships
Administration Timing
The purpose of PEG-MGF in a research context dictates when it should be administered:
- Post-exercise administration: The most studied timing window, typically within 30 minutes to 2 hours after the training session when satellite cell receptivity is highest due to the mechanical damage stimulus already being present
- Rest day administration: Because PEG-MGF's extended half-life means it remains active for days, some protocols administer it on rest days to study continuous satellite cell activation independent of exercise timing
- Injury recovery protocols: In tissue repair research, PEG-MGF is administered immediately following induced tissue damage and continued on a weekly schedule to study accelerated healing
Injection Route
PEG-MGF is most commonly administered via subcutaneous injection in research settings. Some protocols use intramuscular injection when targeting a specific muscle group for localized study, though PEG-MGF's systemic half-life means the injection site is less critical than it would be with unmodified MGF.
Frequency and Cycling
Given its multi-day half-life, PEG-MGF is typically administered 1 to 2 times per week in most research protocols. This dosing frequency maintains consistent satellite cell activation without receptor oversaturation.
Research cycles typically run 4 to 6 weeks, followed by a break period. This cycling approach allows researchers to study discrete phases of muscle adaptation and prevents receptor downregulation that could occur with continuous, indefinite administration.
Reconstitution and Storage
PEG-MGF is supplied as a lyophilized powder and is reconstituted with bacteriostatic water for research use. Once reconstituted, it should be stored refrigerated at 2 to 8 degrees Celsius and protected from light exposure. The PEG coating improves stability compared to unmodified MGF, but proper cold storage remains essential for maintaining peptide integrity.
For accurate reconstitution and dosing calculations, our Peptide Calculator Guide walks through the full process step by step.
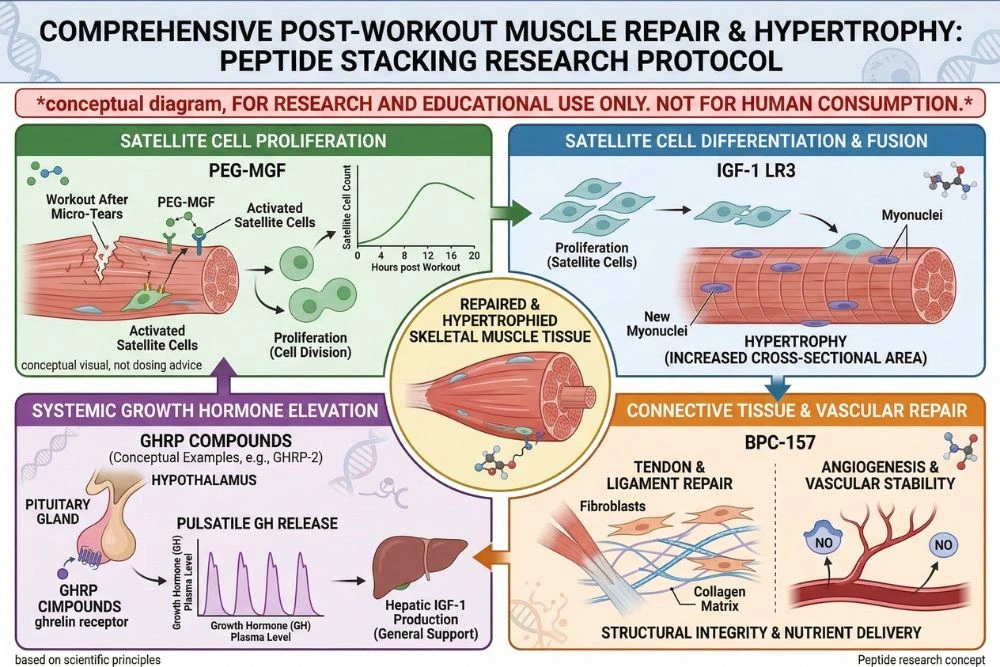
Stacking PEG-MGF with Other Research Peptides
PEG-MGF's unique position in the muscle repair cascade makes it highly complementary to several other research peptides. Strategic combinations allow researchers to study the full arc of muscle growth from initial repair signaling through to full hypertrophic adaptation.
PEG-MGF plus IGF-1 LR3: The Complete Growth Stack
This is the most studied and scientifically logical combination in muscle hypertrophy research. PEG-MGF handles the first phase: activating and proliferating the satellite cell pool. IGF-1 LR3 then handles the second phase: driving those satellite cells to differentiate into mature muscle fibers and fusing them into the existing muscle tissue.
Because these two compounds operate on different receptors and different phases of the repair cascade, their effects are additive. Research protocols using both compounds together have shown greater muscle cross-sectional area increases than either compound alone in animal models.
Timing matters for this combination. PEG-MGF is typically administered post-workout to capture the exercise-induced window of satellite cell receptivity. IGF-1 LR3 can be administered in a separate window, often the following morning, to drive the differentiation phase after proliferation has been initiated.
PEG-MGF plus BPC-157: Recovery and Structural Repair
BPC-157 works through completely different mechanisms from PEG-MGF, primarily targeting blood vessel formation (angiogenesis), collagen synthesis, and systemic anti-inflammatory signaling. Adding BPC-157 to a PEG-MGF protocol addresses a complementary dimension of recovery: the connective tissue and vascular infrastructure that supports the muscle cells PEG-MGF is rebuilding.
For a full breakdown of BPC-157's repair mechanisms, see our guide on BPC-157 Peptide: A Comprehensive Research Guide to Tissue Repair and Recovery.
PEG-MGF plus GHRPs: Growth Hormone Amplification
Combining PEG-MGF with a GHRP like hexarelin or GHRP-2 adds a third layer to the research stack. The GHRP drives a significant GH pulse, which elevates natural IGF-1 from the liver and creates a systemically anabolic environment. PEG-MGF simultaneously activates the local satellite cell population in muscle tissue. Together, the systemic GH/IGF-1 environment and the targeted satellite cell activation provide a comprehensive anabolic and repair stimulus.
For detailed context on how GHRPs fit into this kind of stacking research, our guide on Hexarelin: The Strongest GHRP for Mass and Strength covers GHRP mechanisms and stacking strategies in depth.
Safety Profile and Research Considerations
PEG-MGF has a generally favorable safety profile based on the available research, but like all biologically active research peptides, it comes with considerations that every researcher needs to understand before designing a protocol.
Reported Observations in Research
In animal models and early human-context research, PEG-MGF has been well-tolerated at standard research doses. The most commonly reported observations include mild injection site reactions in some subjects, transient fatigue in the early days following administration as the body responds to elevated satellite cell activity, and in some cases, mild water retention similar to what is seen with other IGF-family peptides.
IGF-1 Pathway Activity and Cell Growth Considerations
Because PEG-MGF activates pathways related to IGF-1 signaling, researchers should be aware of the theoretical considerations associated with growth factor activity. IGF-1-related pathways promote cell proliferation broadly. Research protocols involving subjects with pre-existing conditions associated with abnormal cell growth should be designed with appropriate controls and monitoring.
This is a standard consideration with all IGF-family research peptides and does not represent a unique risk specific to PEG-MGF, but it is an important variable to account for in study design.
PEG Coating Considerations
PEGylation is a well-established and widely used pharmaceutical modification. PEG itself is considered biologically inert and is used in numerous FDA-approved medications. However, some research has noted that very long-term, high-dose PEG exposure can, in rare cases, trigger PEG-specific antibody formation. This is generally not a concern in typical research cycling protocols but is worth noting for study designs involving extended administration periods.
Interaction with Hormonal Systems
PEG-MGF operates primarily through muscle-specific MGF receptors and does not directly interact with pituitary hormones, sex hormones, or the HPG axis. This makes it a relatively clean compound from a hormonal interference standpoint, which is an advantage in research protocols where maintaining stable hormonal backgrounds is important for data quality.
What Makes PEG-MGF Different from Other Muscle-Building Peptides?
PEG-MGF occupies a unique niche in the peptide research landscape because its mechanism is fundamentally different from the other major muscle-building peptides.
PEG-MGF vs. IGF-1 LR3
IGF-1 LR3 drives protein synthesis and satellite cell differentiation through the IGF-1 receptor. It is excellent at the second phase of muscle growth but less effective at the critical first step of pulling dormant satellite cells out of quiescence. PEG-MGF excels at exactly that first step. The two compounds are complementary, not competitive.
PEG-MGF vs. GHRPs
GHRPs like hexarelin and ipamorelin work upstream by stimulating GH release from the pituitary, which then elevates systemic IGF-1. This is a top-down, systemic approach to promoting muscle growth. PEG-MGF works bottom-up, directly at the muscle fiber level through satellite cells. Again, these are complementary approaches that address different parts of the growth equation.
PEG-MGF vs. Follistatin and Myostatin Inhibitors
Myostatin is a natural brake on muscle growth, and myostatin inhibition research aims to remove that brake entirely. PEG-MGF does not inhibit myostatin. Instead, it pushes the accelerator on the satellite cell response. Combining myostatin inhibition with PEG-MGF represents one of the most aggressive approaches in muscle hypertrophy research, removing the growth limit while simultaneously maximizing the repair response.
The specificity of PEG-MGF's mechanism is its greatest research advantage. It does one thing exceptionally well: it activates the cellular machinery that rebuilds damaged muscle. Every researcher studying hypertrophy, recovery, and muscle aging needs this mechanism in their toolkit.
Sourcing Quality PEG-MGF for Research
PEG-MGF synthesis is more complex than many simpler peptides because the PEGylation step must be performed correctly to produce a compound with the intended biological activity. Quality control is critical.
What to Look for in a Supplier
Any PEG-MGF purchased for research should come with a third-party Certificate of Analysis confirming:
- HPLC purity at or above 98 percent
- Mass spectrometry confirming the correct molecular weight including the PEG chain
- Sequence verification of the MGF Ec peptide region
- Batch documentation from an accredited third-party laboratory
Before purchasing any research peptide, our guide on How to Read a Peptide Certificate of Analysis will show you exactly what to look for and which sections matter most for verifying compound quality.
Why PEGylation Quality Matters
Incorrect PEGylation can attach the PEG chain to the wrong position on the peptide, blocking the active Ec region and producing a compound with drastically reduced biological activity. A batch that tests well for purity but has incorrect PEG attachment may still fail to activate satellite cells effectively. Always prioritize suppliers who use advanced mass spectrometry to confirm PEG site specificity, not just total molecular weight.
Recommended Research Supplier
Peptides Finder connects researchers with vetted suppliers who provide full third-party documentation for PEG-MGF and all major research peptides.
Browse PEG-MGF Research Compounds at Peptides Finder
We may earn a commission from purchases made through links on this page.
Frequently Asked Questions
What does PEG-MGF stand for?
PEG-MGF stands for Polyethylene Glycol-Mechano Growth Factor. MGF is a splice variant of IGF-1 that the body produces naturally in response to mechanical stress on muscle tissue, such as from resistance training. The PEG prefix refers to the polyethylene glycol chain that has been chemically attached to the MGF peptide. This PEGylation process extends the peptide's half-life from just a few minutes to approximately 48 to 72 hours, allowing it to circulate systemically and reach multiple muscle groups rather than breaking down before it can do its job.
How does PEG-MGF repair muscle tissue?
PEG-MGF repairs muscle tissue by activating satellite cells, which are the adult stem cells that live in a dormant state alongside muscle fibers. The unique Ec peptide sequence at the C-terminal end of MGF binds to receptors on satellite cells and triggers them to wake up, multiply, migrate to sites of muscle damage, and mature into myoblasts that fuse with damaged fibers or form new ones. This satellite cell activation is the fundamental mechanism of muscle tissue repair after training, and PEG-MGF amplifies this natural process by providing a stronger and more sustained activation signal than exercise alone generates.
What is the difference between PEG-MGF and regular MGF?
The core biological difference is half-life and reach. Unmodified MGF has a half-life of just a few minutes in the bloodstream, making it a local signal that works only on the tissue immediately surrounding the injection site before it is broken down. PEG-MGF has a half-life of 48 to 72 hours and circulates systemically, reaching muscle tissue throughout the body. For research purposes, unmodified MGF is used when studying localized, immediate effects on a specific muscle site. PEG-MGF is used when systemic satellite cell activation, whole-body recovery, or sustained multi-day signaling is the research objective.
Can PEG-MGF be stacked with IGF-1 LR3?
Yes, and this is one of the most scientifically logical combinations in muscle hypertrophy research. PEG-MGF and IGF-1 LR3 work in sequence on the same biological process. PEG-MGF activates and proliferates the satellite cell pool during the first phase of muscle repair. IGF-1 LR3 then drives those satellite cells to differentiate into mature muscle cells during the second phase. Because they operate on different receptors and different phases of the repair cascade, their effects are additive rather than redundant, and published animal research consistently shows greater muscle mass gains from the combination than from either compound alone.
How often should PEG-MGF be administered in research protocols?
Because PEG-MGF has an extended half-life of approximately 48 to 72 hours, research protocols typically administer it one to two times per week. More frequent administration risks receptor oversaturation and provides no meaningful increase in biological activity given how long each dose remains active. The most studied timing is post-exercise, administered within 30 minutes to 2 hours after training to capture the window of peak satellite cell receptivity. For injury recovery research, administration timing is centered around the acute damage event rather than an exercise schedule.
What are the main research applications of PEG-MGF?
The primary research applications of PEG-MGF fall into four categories. First, post-workout muscle repair research, studying how accelerated satellite cell activation affects recovery speed and training adaptation. Second, muscle hypertrophy research, studying how expanded satellite cell populations contribute to greater muscle cross-sectional area growth over time. Third, sarcopenia research, studying whether exogenous MGF supplementation can restore blunted satellite cell responses in aging muscle tissue. Fourth, muscle preservation research, studying how MGF signaling protects against atrophy during periods of immobilization or injury-forced inactivity.
How is PEG-MGF different from growth hormone and GHRPs?
Growth hormone and GHRPs work systemically from the top down. They stimulate the pituitary and liver to create an anabolic hormonal environment that benefits muscle tissue indirectly. PEG-MGF works locally from the bottom up, acting directly on satellite cells within muscle tissue through a receptor that is specific to MGF. This means PEG-MGF bypasses the hormonal axis entirely and delivers its growth signal directly to the cellular machinery that does the actual repair work. The two approaches target completely different parts of the muscle growth equation and are highly complementary when combined in research protocols.
Conclusion: PEG-MGF as the Targeted Tool for Muscle Repair Research
Most peptides in muscle research work indirectly, creating a hormonal environment that the body then translates into muscle growth over time. PEG-MGF is different. It goes straight to the source, directly activating the satellite cells that physically rebuild damaged muscle fibers.
This specificity is what makes PEG-MGF so valuable. Researchers do not have to hope that a GH spike or an IGF-1 elevation eventually reaches the right cells at the right time. PEG-MGF delivers the activation signal directly to the cellular workforce that does the rebuilding. The PEG modification solves the one problem that made natural MGF impractical for systemic research: it gives the peptide time to actually do its job.
From post-workout hypertrophy research to sarcopenia science to injury recovery studies, PEG-MGF offers a level of mechanistic precision that few other compounds can match. When combined intelligently with IGF-1 LR3, BPC-157, or GHRPs, it becomes part of a comprehensive research stack that covers muscle repair from every angle.
For any researcher serious about understanding how muscle grows, recovers, and ages, PEG-MGF belongs at the center of the conversation.
Explore the full library of muscle repair and growth peptide research guides at Peptides Finder, and stay current with the latest developments in regenerative muscle science.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. PEG-MGF is sold as a research chemical. Research peptides are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any research compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.