Tesamorelin: The FDA-Approved Peptide Targeting Visceral Belly Fat
Discover how tesamorelin targets visceral belly fat through GH release. FDA-approved GHRH analog. Learn dosing protocols, clinical evidence & results.
There's a reason most diets fail at the final hurdle.
You can count every calorie, crush every workout, and still have that stubborn layer of fat clinging to your midsection. It's not a failure of willpower. It's biology.
The fat wrapped around your internal organs (called visceral adipose tissue) doesn't respond to traditional diet and exercise the way subcutaneous fat does. It's metabolically active, hormonally resistant, and dangerously linked to diabetes, heart disease, and insulin resistance.
Enter tesamorelin.
This is not another unproven research compound. Tesamorelin is an FDA-approved synthetic peptide analog of growth hormone-releasing hormone (GHRH), specifically designed to reduce visceral fat accumulation in patients with HIV-associated lipodystrophy. But its clinical success has sparked intense research interest in metabolic health, body recomposition, and longevity medicine.
This guide explains how tesamorelin works at the molecular level, why it selectively targets dangerous belly fat, the clinical evidence supporting its use, and what researchers need to know about dosing, stacking, and safety protocols in 2026.
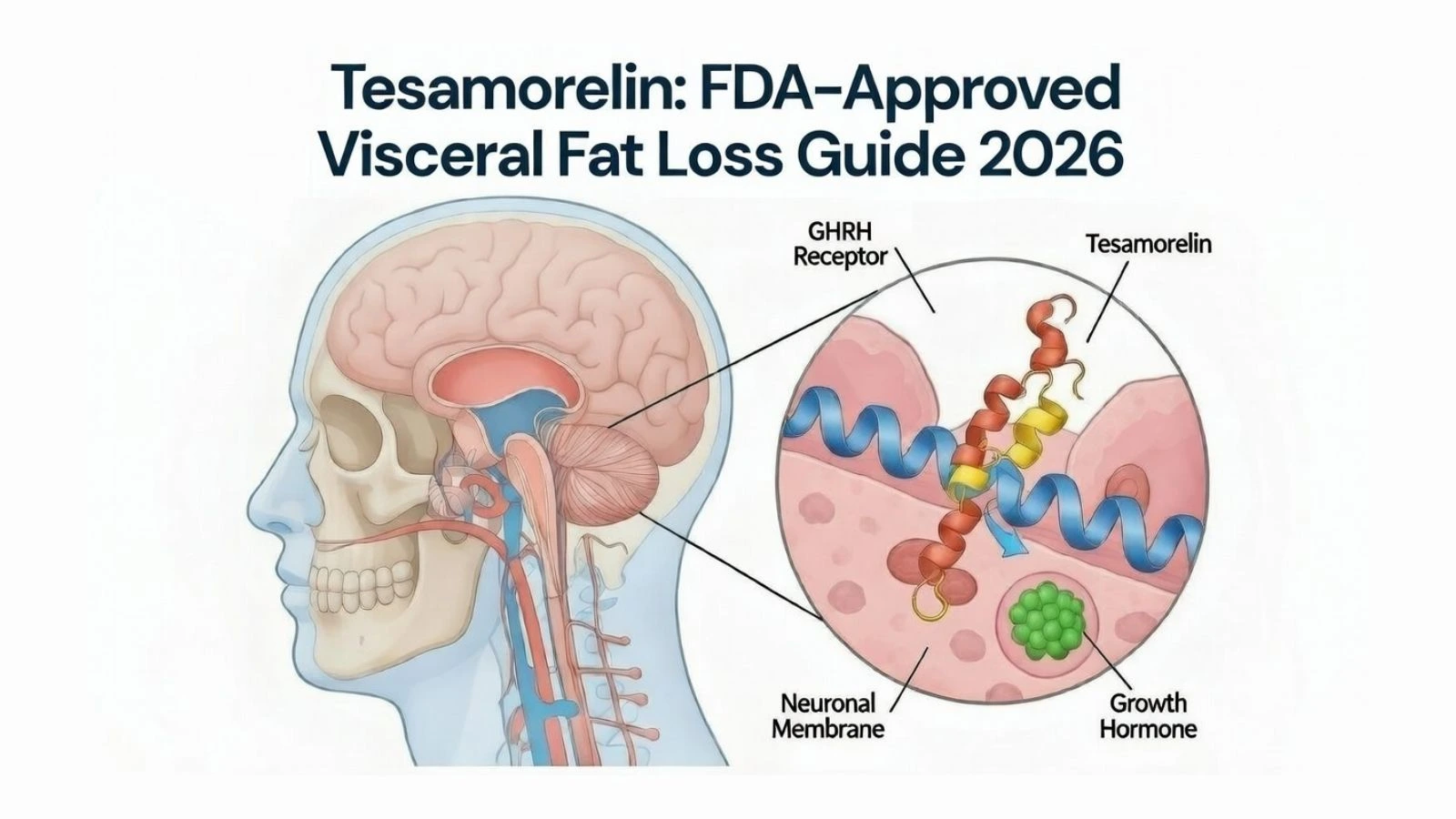
What Is Tesamorelin? The Modified GHRH Analog
To understand tesamorelin, you first need to understand the natural hormone it mimics.
Growth hormone-releasing hormone (GHRH) is a 44-amino acid peptide produced by the hypothalamus. Its job is simple but critical: signal the anterior pituitary gland to synthesize and secrete growth hormone (GH).
The problem? Natural GHRH has a half-life measured in minutes. It gets chewed up by an enzyme called dipeptidyl peptidase-4 (DPP-4) almost immediately after release.
Tesamorelin solves this problem through a single, elegant modification: the addition of a trans-3-hexenoic acid group to the N-terminus of the peptide chain. According to the FDA's chemistry review, this structural change makes tesamorelin resistant to enzymatic degradation while preserving its ability to bind to GHRH receptors.
The result is a peptide that:
- Maintains full biological activity
- Has significantly extended stability in circulation
- Triggers pulsatile GH release that mimics natural rhythms
- Preserves the body's endogenous feedback mechanisms
Unlike synthetic growth hormone injections (which flood the system with exogenous hormone and can suppress natural production), tesamorelin works with your body's own regulatory systems.
Want to understand the broader landscape of growth hormone peptides? Read our guide on Peptides for Fat Loss: The Science of Metabolic Optimization and Lipolysis.
The Mechanism: How Tesamorelin Targets Visceral Fat
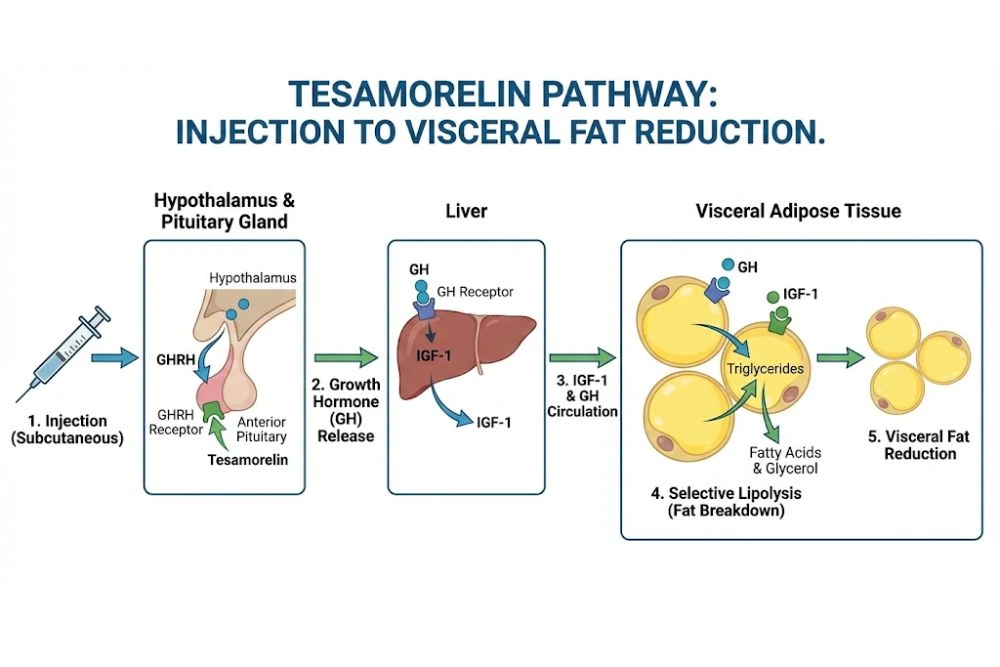
The story of how tesamorelin reduces belly fat is a cascade of hormonal signaling that starts in the brain and ends at the fat cell.
Step 1: Receptor Binding in the Pituitary
After subcutaneous injection, tesamorelin enters systemic circulation and crosses the blood-brain barrier to reach the anterior pituitary gland. There, it binds to GHRH receptors on somatotroph cells (specialized cells responsible for growth hormone synthesis).
This binding activates adenylyl cyclase, triggering an intracellular signaling cascade that culminates in the transcription of growth hormone genes and the secretion of GH into the bloodstream.
Step 2: Elevation of IGF-1
Growth hormone doesn't work alone. Once released, it travels to the liver and stimulates the production of insulin-like growth factor 1 (IGF-1), the primary mediator of GH's anabolic and metabolic effects.
Clinical trials published in the New England Journal of Medicine show that tesamorelin can raise IGF-1 levels significantly within weeks of starting treatment. This isn't a small bump, it's a complete metabolic shift.
Step 3: Selective Lipolysis of Visceral Fat
Here's where it gets interesting.
Growth hormone and IGF-1 promote lipolysis (the breakdown of stored triglycerides into free fatty acids). But tesamorelin doesn't just burn fat indiscriminately. It shows a marked preference for visceral adipose tissue.
The prevailing theory involves insulin sensitivity. Visceral fat is highly sensitive to insulin, and elevated insulin levels promote fat storage in this depot. By improving insulin sensitivity (a documented effect of GH), tesamorelin may reduce the hormonal signal that locks visceral fat in place.
In two landmark Phase III clinical trials published on PubMed, participants treated with tesamorelin for 26 weeks experienced:
- 10.9% reduction in visceral adipose tissue (measured by CT scan)
- Significant decreases in waist circumference and waist-to-hip ratio
- Minimal changes in total body weight (fat loss, not muscle wasting)
A separate study in JAMA found even more impressive results: a mean reduction of 34 cm² in visceral adipose tissue compared to placebo, along with modest reductions in liver fat.
This selectivity is what makes tesamorelin clinically valuable. You're not just losing weight, you're losing the dangerous fat.
Why Visceral Fat Matters More Than You Think
Not all fat is created equal.
Subcutaneous fat (the kind you can pinch) sits just beneath the skin. It's relatively benign from a metabolic perspective. Visceral fat, on the other hand, wraps around your liver, pancreas, intestines, and other vital organs. It's metabolically active, secreting inflammatory cytokines and hormones that disrupt insulin signaling, promote atherosclerosis, and increase cardiovascular risk.
Studies have shown that visceral adiposity is independently associated with:
- Type 2 diabetes
- Cardiovascular disease
- Non-alcoholic fatty liver disease (NAFLD)
- Certain cancers
- Cognitive decline
This is why tesamorelin's FDA approval in 2010 was so significant. It wasn't approved for cosmetic fat loss. It was approved because reducing visceral fat in HIV patients with lipodystrophy improved their metabolic health markers and reduced long-term disease risk.
Curious about other approaches to metabolic optimization? Check out our article on GLP-1 Drugs Explained: The Science Behind the Weight Loss Revolution.
Clinical Applications: FDA-Approved and Off-Label Use
FDA-Approved Use: HIV-Associated Lipodystrophy
Tesamorelin received FDA approval in 2010 under the brand name Egrifta (later reformulated as Egrifta SV) for the treatment of excess abdominal fat in HIV-positive patients on highly active antiretroviral therapy (HAART).
HAART is life-saving, but it comes with a metabolic cost: lipodystrophy. This is abnormal fat redistribution characterized by loss of fat in the face and limbs and accumulation in the abdomen. This isn't just cosmetic. Patients with HIV lipodystrophy face increased cardiovascular risk, insulin resistance, and psychological distress.
Tesamorelin addresses this by reducing the visceral fat depot without worsening peripheral fat loss.
Off-Label Interest: Metabolic Syndrome and Body Recomposition
While tesamorelin is only FDA-approved for HIV lipodystrophy, physicians may prescribe it off-label for other conditions at their discretion. Research is exploring its potential in:
- Non-alcoholic fatty liver disease (NAFLD)
- Metabolic syndrome
- Age-related growth hormone deficiency
- Cognitive impairment (preliminary studies suggest potential benefits)
It's also garnered attention in bodybuilding and physique enhancement circles for its ability to reduce stubborn abdominal fat while preserving lean mass, though this use remains off-label and should only be pursued under medical supervision.
Interested in comprehensive fat loss research? See our guide Peptides for Fat Loss: The Science of Metabolic Optimization and Lipolysis.
Tesamorelin Dosing and Administration Protocols
Precision matters when working with growth hormone-releasing peptides.
Standard Clinical Dosing
The FDA-approved dose for tesamorelin is 2 mg once daily, administered via subcutaneous injection into the abdominal area.
Clinical trials used this exact protocol:
- Timing: Evening injection (aligns with natural nocturnal GH pulse)
- Frequency: Daily, at least 2 hours after last meal
- Injection site: Rotate within abdominal region to prevent lipohypertrophy
Reconstitution: The Wall Method
Tesamorelin arrives as a lyophilized powder that must be reconstituted with sterile water or bacteriostatic water before injection.
Follow the "Wall Method" to preserve peptide integrity:
- Draw the appropriate volume of bacteriostatic water into an insulin syringe.
- Insert the needle into the tesamorelin vial at a 45-degree angle.
- Aim the needle tip at the inside wall of the glass vial.
- Gently push the plunger so water trickles down the wall (never spray directly onto the powder).
- Gently swirl (do NOT shake) until the solution is clear.
Need help calculating exact doses? Use our Peptide Calculator for Accurate Research to ensure precision.
Video: What Is Tesamorelin? How It Works + What To Expect
Watch this comprehensive breakdown by Josh Holyfield explaining tesamorelin's mechanism of action, expected results, and clinical applications.
Cycle Length and Blood Monitoring
Clinical studies evaluated tesamorelin over 26-week cycles, but some researchers and physicians extend use for 6 to 12 months or longer under close supervision.
Recommended monitoring:
- Baseline IGF-1 levels before starting
- IGF-1 monitoring every 4 to 8 weeks during treatment
- Fasting glucose and HbA1c (tesamorelin can affect insulin sensitivity)
- Liver function tests (especially if using for NAFLD research)
Some protocols include off cycles every 12 to 16 weeks to prevent receptor desensitization, though clinical data on this is limited.
Peptide Stacking: Synergistic Combinations with Tesamorelin
Many researchers and physicians combine tesamorelin with other growth hormone secretagogues to amplify GH release through complementary pathways.
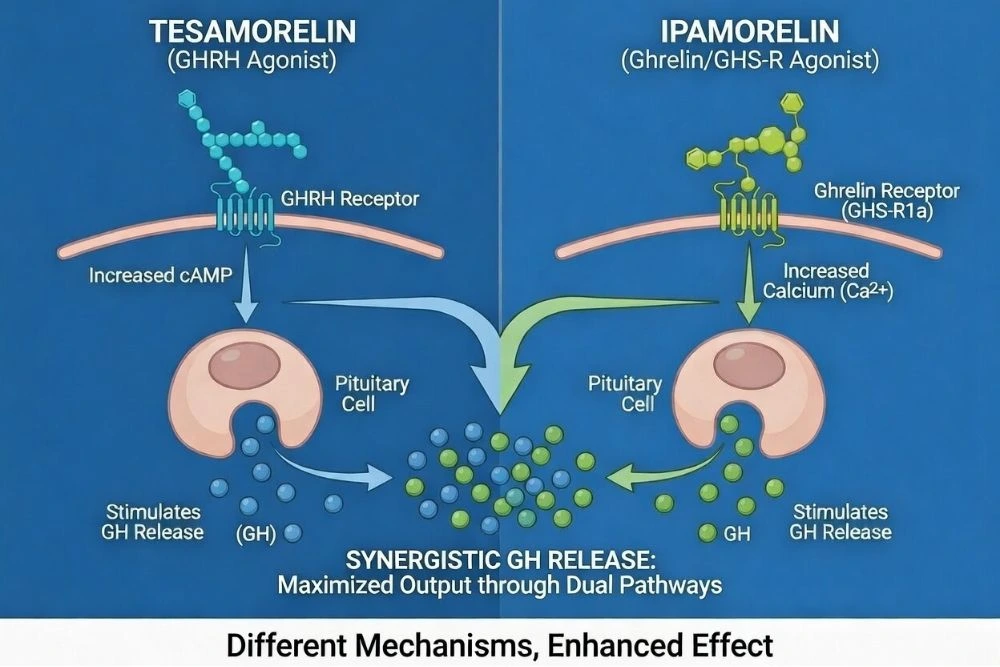
Tesamorelin + Ipamorelin
This is one of the most popular stacks.
Ipamorelin is a growth hormone-releasing peptide (GHRP) that works by mimicking ghrelin and blocking somatostatin, the hormone that inhibits GH release. Tesamorelin, as a GHRH analog, stimulates GH production directly.
Together, they create a "two-pronged" effect:
- Ipamorelin removes the brake (somatostatin inhibition).
- Tesamorelin presses the accelerator (GHRH stimulation).
The result is a more pronounced GH pulse without the cortisol and prolactin spikes associated with older GHRPs like GHRP-6.
For a detailed comparison of GH secretagogues, read CJC-1295 vs. Ipamorelin: Which Growth Hormone Secretagogue is Best?.
Tesamorelin + CJC-1295 (No DAC)
CJC-1295 without the Drug Affinity Complex (DAC) is another GHRH analog with a slightly different pharmacokinetic profile than tesamorelin. Some protocols use both to extend the GH release window throughout the day.
However, stacking two GHRH analogs may offer diminishing returns since they act on the same receptor. Most experts recommend pairing tesamorelin with a GHRP (like ipamorelin) rather than another GHRH.
Side Effects and Safety Considerations
Tesamorelin has been extensively studied in clinical trials involving over 800 participants. While generally well-tolerated, it's not without potential side effects.
Common Side Effects (Mild and Manageable)
- Injection site reactions: Redness, itching, or mild swelling (most common)
- Arthralgia: Joint pain or stiffness (usually resolves with continued use)
- Peripheral edema: Mild fluid retention in hands or feet
- Muscle pain: Transient myalgia
In clinical trials, about 5% of participants discontinued tesamorelin due to adverse events, most commonly joint pain or injection site reactions.
Serious but Rare Side Effects
- Glucose intolerance: Tesamorelin can increase blood sugar levels. Not recommended for individuals with uncontrolled diabetes.
- Hypersensitivity reactions: Rare allergic reactions (rash, difficulty breathing).
- Tumor growth concerns: Theoretical risk of stimulating pre-existing tumors (contraindicated in active malignancy).
Contraindications
Do NOT use tesamorelin if you have:
- Active malignancy or history of cancer
- Pituitary gland tumors or recent pituitary surgery
- Pregnancy or breastfeeding
- Severe diabetic retinopathy
Always verify purity before use. Learn how to read Scientific Peptides: How to Read a Certificate of Analysis (CoA).
Storage and Stability: Protecting Your Investment
Peptides are fragile molecules. Improper storage will destroy tesamorelin's biological activity long before you inject it.
Lyophilized Powder (Before Reconstitution)
- Store in freezer (-20°C or colder).
- Protect from light.
- Can remain stable for 24+ months when properly stored.
Reconstituted Solution
Once mixed with bacteriostatic water:
- Store in refrigerator (2 to 8°C).
- Use within 2 to 4 weeks maximum.
- Never freeze reconstituted peptides.
- Protect from direct sunlight.
If your solution turns cloudy or develops particles, discard it immediately. The peptide has degraded.

Research-Grade vs. Pharmaceutical-Grade Tesamorelin
This distinction is critical and often misunderstood.
Pharmaceutical-grade tesamorelin (brand name Egrifta) is FDA-approved, manufactured under strict cGMP (current Good Manufacturing Practice) guidelines, and available only by prescription. Every vial is rigorously tested for purity, sterility, and potency.
Research-grade tesamorelin is sold online as a "research chemical" with a disclaimer that it is "not for human consumption." These products:
- May contain impurities, degradation products, or incorrect peptide sequences
- Are not subject to FDA oversight
- Often lack third-party testing or valid certificates of analysis
- Carry serious safety and legal risks when used off-label
If you're considering tesamorelin for research purposes, always source from reputable vendors who provide third-party testing via HPLC (high-performance liquid chromatography) and mass spectrometry.
Frequently Asked Questions
How long does it take to see results with tesamorelin?
Clinical trials showed measurable reductions in visceral fat within 6 to 12 weeks of daily use. However, some metabolic benefits (improved lipid profiles, insulin sensitivity) may appear earlier. Full body recomposition effects typically require 3 to 6 months of consistent use.
Can women use tesamorelin?
Yes. Clinical trials included both men and women, and efficacy was demonstrated in both sexes. However, tesamorelin is contraindicated during pregnancy and breastfeeding due to potential effects on the fetus and infant.
Does tesamorelin build muscle?
Tesamorelin's primary effect is fat reduction, not direct muscle hypertrophy. However, by elevating GH and IGF-1, it creates a more anabolic hormonal environment that may support muscle recovery and protein synthesis when combined with resistance training. It's best thought of as a body recomposition tool rather than a standalone muscle builder. For muscle-building research, see Peptides for Muscle Growth: The Ultimate Stacking Guide
What's the difference between tesamorelin and sermorelin?
Both are GHRH analogs, but tesamorelin has a longer half-life and greater stability due to its N-terminal modification. Sermorelin is a shorter peptide (29 amino acids) and is metabolized more quickly. Tesamorelin has more robust clinical evidence for visceral fat reduction.
Is tesamorelin safe for long-term use?
Clinical trials evaluated tesamorelin for up to 52 weeks. Long-term safety data beyond one year is limited. Most adverse events are mild and reversible upon discontinuation. Ongoing medical supervision and blood monitoring are essential for extended use.
Conclusion: The Precision Tool for Stubborn Fat
In the evolving landscape of metabolic health and body recomposition, tesamorelin stands out not for hype, but for evidence.
It's one of the few peptides that has:
- Survived rigorous FDA scrutiny
- Demonstrated reproducible results in placebo-controlled trials
- Shown selective targeting of the most dangerous type of fat
- Preserved natural hormonal feedback systems
Whether you're researching metabolic dysfunction, exploring body recomposition protocols, or investigating longevity medicine, tesamorelin offers a scientifically validated pathway to reducing visceral adiposity.
But remember: precision matters. Proper reconstitution, accurate dosing, consistent injection protocols, and regular blood monitoring aren't optional. They're essential for both safety and efficacy.
As research continues to uncover the full metabolic and longevity benefits of growth hormone optimization, tesamorelin remains a foundational tool in the peptide therapy toolkit.
Official Medical Disclaimer
The information provided in this guide is for informational and educational purposes only.
Peptides and GLP-1 research compounds are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in a controlled environment. The statements regarding these products have not been evaluated by the Food and Drug Administration (FDA). Always consult your local laws and institutional guidelines regarding the use of peptides in research. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.