CJC-1295 vs. Ipamorelin: Which Growth Hormone Secretagogue is Best?
CJC-1295 vs. ipamorelin: discover how these two growth hormone secretagogues work, why researchers combine them, and what the clinical evidence actually shows.
CJC-1295 vs. Ipamorelin: Mechanisms, Research, and Why They Are Often Combined
When researchers study growth hormone secretagogues, two compounds keep appearing together in protocols, literature, and discussions: CJC-1295 and ipamorelin. On the surface, they look similar. Both stimulate growth hormone release. Both are injectable peptides. Both are studied in contexts ranging from metabolic health to body composition and recovery. But the way they work is fundamentally different, and understanding that difference is essential for anyone seriously evaluating these compounds.
This guide breaks down what each compound is, how each mechanism functions, what the clinical research has found, why they are so often combined, and what researchers need to consider when working with either or both. The short answer to the question in the title is that it is the wrong question. For most research purposes, CJC-1295 and ipamorelin are not alternatives to each other. They are partners.
Understanding the Growth Hormone Axis: The Foundation for Everything
To make sense of CJC-1295 and ipamorelin, you need a working understanding of how the body controls growth hormone secretion, because each compound targets a different part of that control system.
Growth hormone is produced by somatotroph cells in the anterior pituitary gland and released in pulses throughout the day, with the largest pulses occurring during deep sleep. The pituitary does not decide on its own when to release GH. It takes instructions from two main inputs, and these inputs originate in the hypothalamus and the peripheral tissues of the body.
The first input is growth hormone-releasing hormone, known as GHRH. This is a 44-amino acid peptide that the hypothalamus releases in pulses. When GHRH binds to its receptor on somatotroph cells, it activates adenylate cyclase, raises intracellular cyclic AMP, and triggers GH synthesis and secretion. GHRH is the primary on-signal for GH release.
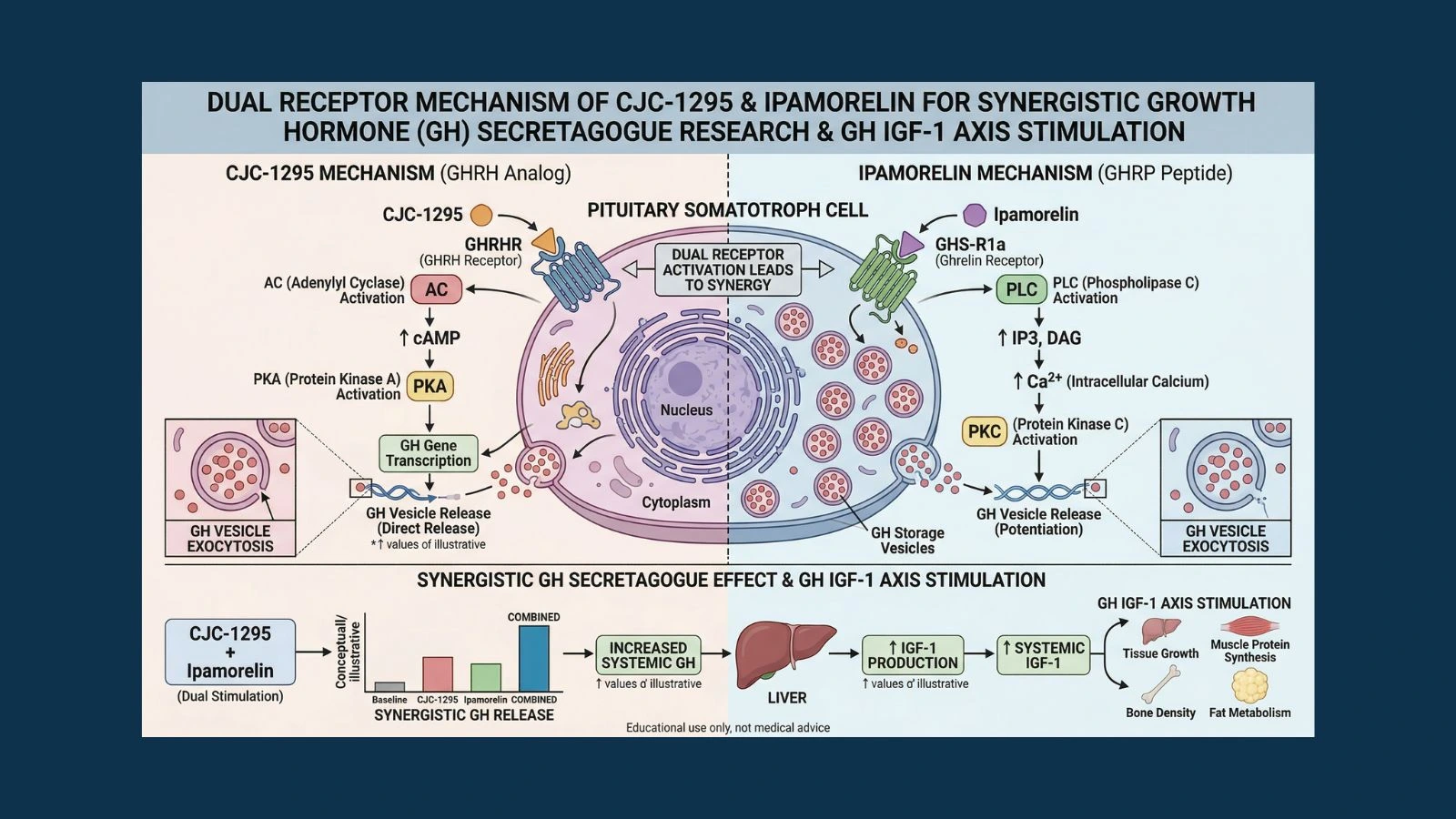
The second input is the ghrelin/GHS-R1a pathway. Ghrelin is a peptide hormone produced mainly in the stomach that acts as a hunger signal, but it also has a powerful GH-releasing effect. It does this by binding to a completely separate receptor on somatotroph cells called the growth hormone secretagogue receptor (GHS-R1a). When this receptor is activated, it uses a different second messenger cascade, one involving phospholipase C and calcium signaling rather than the cAMP pathway used by GHRH.
These two receptor systems work on the same cell but through independent molecular pathways. The most important implication of this is that stimulating both receptors simultaneously produces a GH response that is significantly greater than the sum of what either can produce alone. This supra-additive synergy was definitively confirmed in human subjects by the foundational research published in PubMed (PMID 2108187), which showed that combined GHRH plus GHRP administration produced synergistic GH release well beyond what each pathway could achieve independently.
CJC-1295 is engineered to act on the GHRH receptor pathway. Ipamorelin is engineered to act on the GHS-R1a pathway. This is why combining them is so mechanistically compelling: they hit different targets on the same cell and their signals amplify each other.
CJC-1295: The Long-Acting GHRH Analog
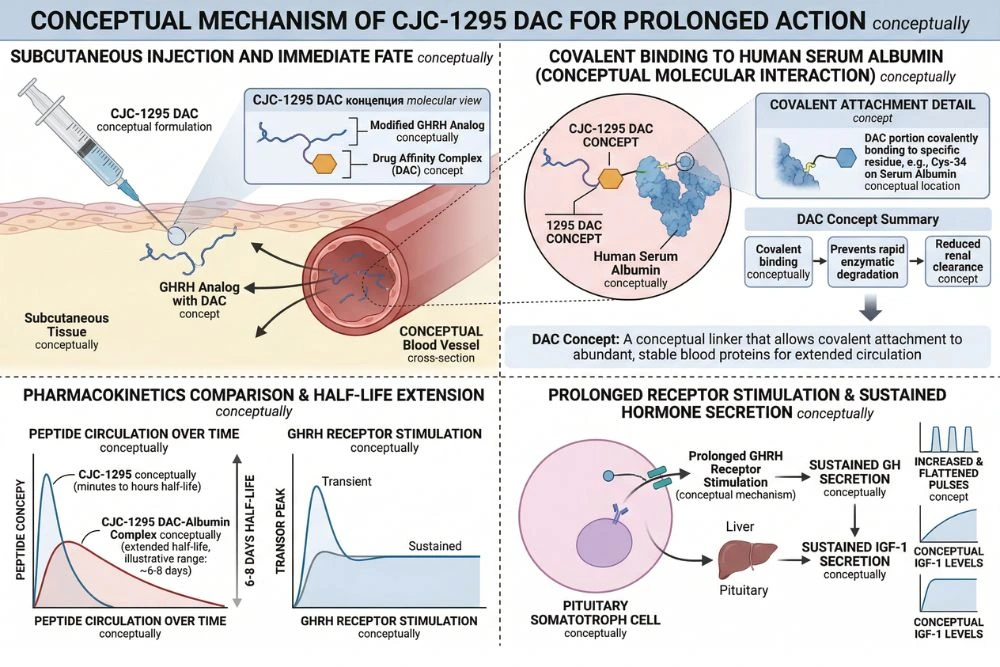
CJC-1295 is a synthetic analog of GHRH, specifically based on the first 29 amino acids of the natural GHRH sequence (GHRH 1-29). Native GHRH has a plasma half-life of just 3 to 7 minutes because it is rapidly cleaved by an enzyme called dipeptidyl peptidase-4 (DPP-4). This short half-life severely limits its therapeutic utility as an injectable compound.
CJC-1295 was engineered to solve this problem in two ways. First, strategic amino acid substitutions were made to the sequence to resist enzymatic degradation. Second, in its most studied form (CJC-1295 with DAC), a Drug Affinity Complex was added that allows the peptide to covalently bind to endogenous serum albumin after injection. Albumin has a naturally long circulating half-life of about 19 days, and anything that binds to it inherits a portion of that stability.
The Landmark Clinical Study
The defining research for CJC-1295 was a randomized, double-blind, placebo-controlled trial published in the Journal of Clinical Endocrinology and Metabolism, available via PubMed (PMID 16352683). The study enrolled healthy adults ages 21 to 61 and tested single and multiple subcutaneous doses of CJC-1295 with DAC.
The results were striking. A single injection produced dose-dependent increases in mean plasma GH concentrations by 2 to 10-fold above baseline values, and these elevated levels were sustained for 6 days or more. Mean plasma IGF-1 concentrations rose by 1.5 to 3-fold and remained elevated for 9 to 11 days. The estimated half-life of CJC-1295 was confirmed at 5.8 to 8.1 days. With multiple doses administered weekly or biweekly, IGF-1 levels remained above baseline for up to 28 days. No serious adverse reactions were reported across the entire trial.
Pulsatility Is Preserved
A critical follow-up study addressed an important mechanistic question: does continuous GHRH stimulation from a long-acting analog flatten out the natural pulsatile rhythm of GH secretion? Pulsatility matters because many of the physiological effects of GH depend on its pulsatile pattern rather than a sustained flat elevation.
The answer, reported in PubMed (PMID 17018654), was that pulsatility is preserved. Even with continuous GHRH receptor stimulation from CJC-1295, GH secretion continued in pulses. What changed was that trough levels between pulses were elevated, meaning the baseline was raised while the pulsatile pattern was maintained. This is an important distinction from exogenous GH administration, which overrides the body's own GH regulation entirely.
CJC-1295 With DAC vs. Without DAC
An important distinction in research with CJC-1295 is whether the DAC modification is present. CJC-1295 without DAC, sometimes called Modified GRF 1-29 or Mod GRF 1-29, has a much shorter half-life of approximately 30 minutes. It produces sharper, more acute GH pulses that more closely mimic the natural GHRH pulse pattern. CJC-1295 with DAC provides the multi-day sustained elevation documented in the clinical trials.
These two forms produce meaningfully different pharmacodynamic profiles and are used to study different research questions. Researchers need to clearly identify which form they are working with, as protocols and expected outcomes differ substantially between them.
Ipamorelin: The Most Selective Growth Hormone Secretagogue
Ipamorelin is a synthetic pentapeptide developed by Novo Nordisk and first described in the scientific literature in 1998. Its full sequence is Aib-His-D-2-Nal-D-Phe-Lys-NH2. It acts as a selective agonist of the GHS-R1a receptor, the same receptor activated by ghrelin, but with a critically different selectivity profile from earlier GHRPs.
The Discovery of Ipamorelin's Selectivity
Before ipamorelin, the available GHRP compounds, including GHRP-2 and GHRP-6, stimulated GH release effectively but also caused unwanted hormonal side effects. Both GHRP-2 and GHRP-6 elevated plasma ACTH and cortisol alongside GH, meaning they activated the stress hormone pathway as a secondary effect. For research protocols lasting weeks or months, chronic cortisol elevation creates significant confounding variables and potential adverse effects.
Ipamorelin changed this. The landmark discovery paper, published by Raun and colleagues at Novo Nordisk and available at PubMed (PMID 9849822), tested ipamorelin head-to-head against GHRP-2 and GHRP-6 in pigs across a range of doses. Ipamorelin matched GHRP-6 in GH-releasing potency and efficacy. None of the GH secretagogues tested affected FSH, LH, prolactin, or TSH. But when it came to ACTH and cortisol, the difference was dramatic: GHRP-2 and GHRP-6 both significantly elevated these stress hormones, while ipamorelin did not raise ACTH or cortisol levels significantly different from those seen with GHRH stimulation alone.
Most importantly, this selectivity was maintained even at doses more than 200 times higher than the GH-releasing ED50. This means ipamorelin does not just have a wider therapeutic window than other GHRPs, it maintains selectivity at doses that would produce substantial stress hormone activation with older compounds. The Novo Nordisk team concluded that ipamorelin was the first GHRP-receptor agonist with a selectivity for GH release comparable to that of GHRH itself.
What Makes Ipamorelin Unique Among GHRPs
Beyond cortisol and ACTH selectivity, ipamorelin also does not significantly raise prolactin or cause the intense appetite stimulation associated with GHRP-6. GHRP-6 activates ghrelin receptor signaling in the hypothalamus in a way that produces strong hunger signals, which is a confounding variable in body composition research. Ipamorelin's more selective receptor binding profile avoids this, making it cleaner from a research perspective when appetite effects are not the variable being studied.
Ipamorelin produces a sharp, rapid GH pulse peaking around 30 to 45 minutes after administration with a duration of 2 to 3 hours. This acute pulse profile makes it highly compatible with CJC-1295 without DAC, which has a similar time course, or with CJC-1295 with DAC as a background elevation booster.
CJC-1295 Plus Ipamorelin: Why Researchers Combine Them
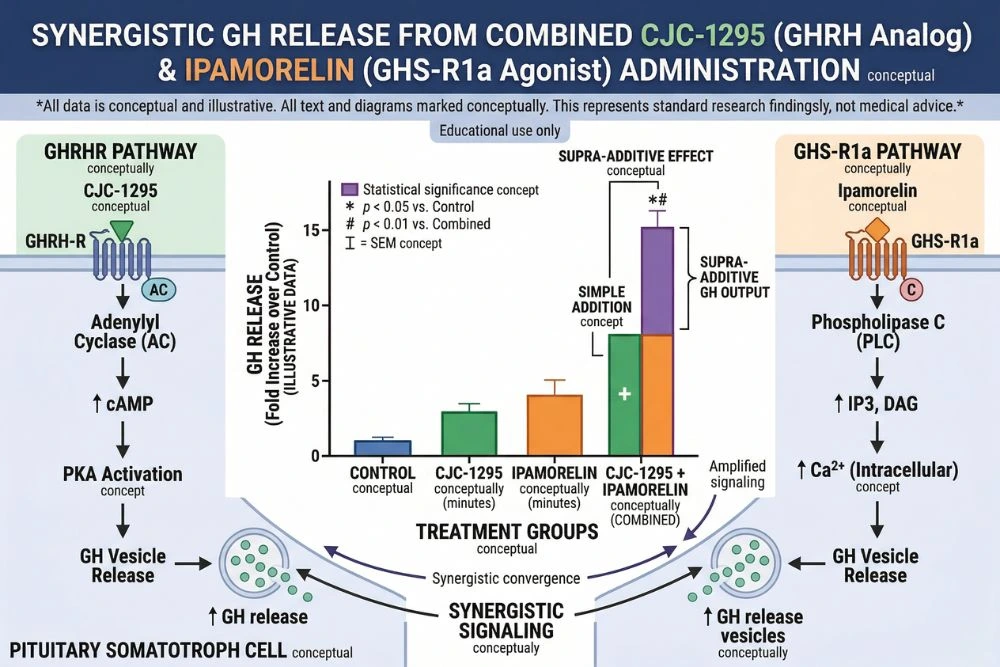
The combination of CJC-1295 and ipamorelin has become one of the most commonly studied secretagogue pairings in research because it directly exploits the dual-receptor architecture of pituitary somatotrophs. CJC-1295 activates the GHRH receptor (cAMP/PKA pathway). Ipamorelin activates the GHS-R1a receptor (PLC/calcium pathway). Together, these two independent signaling cascades converge on the somatotroph to produce a GH release greater than either compound could stimulate alone.
This is not merely an additive effect. The GHRH+GHRP synergy is a well-characterized pharmacological phenomenon with a clear mechanistic basis. When both pathways are stimulated simultaneously, the two second-messenger systems reinforce each other at the level of GH exocytosis. The research established in PubMed (PMID 2108187) demonstrated that sub-maximal doses of GHRP plus GHRH produced GH release synergistically in normal men, with the combined response exceeding what would be expected from simple addition of the individual responses.
What Each Compound Contributes to the Combination
In a typical combined research protocol, each compound brings distinct and complementary properties:
- CJC-1295 with DAC: Provides the sustained baseline elevation of GH and IGF-1 across days. A single injection per week maintains elevated trough GH levels between pulses, raises mean 24-hour GH output, and produces the durable IGF-1 elevation that drives many downstream anabolic and reparative effects.
- Ipamorelin: Provides sharp, clean pulsatile GH bursts at the time of injection through GHS-R1a activation, without confounding cortisol or prolactin signals that would complicate interpretation of results. Its GH pulses add amplitude on top of the baseline elevation established by CJC-1295.
- The combined result: Higher peak GH pulses than ipamorelin alone, higher baseline GH and IGF-1 than would be achieved by CJC-1295 alone, and a more physiologically complete GH secretion pattern that engages both the GHRH and ghrelin receptor systems the way natural GH regulation does.
Maintaining Physiological Feedback Mechanisms
An important safety feature of both CJC-1295 and ipamorelin is that neither bypasses the pituitary's own negative feedback system. Both compounds stimulate the pituitary to secrete its own GH rather than supplying exogenous GH directly. This means the hypothalamic feedback loop, including somatostatin release in response to elevated GH, remains functional. The result is a GH output that, while amplified, cannot escalate to supraphysiological levels the way exogenous GH injections can. This self-limiting property is a significant advantage for research protocols studying sustained GH axis stimulation.
The proteomic analysis published via PMC (PMC2787983), which tracked serum protein changes in healthy men after CJC-1295 administration, provides biomarker evidence for the downstream consequences of GH and IGF-1 elevation through this pathway, including changes in apolipoprotein and transthyretin isoforms consistent with GH axis activation.
Key Research Applications for CJC-1295 and Ipamorelin
The clinical and preclinical literature points toward several primary areas where GH secretagogue research with CJC-1295 and ipamorelin has been most active.
Body Composition Research
GH and IGF-1 are powerful regulators of body composition. GH promotes lipolysis in adipose tissue and protein synthesis in muscle. IGF-1 amplifies the anabolic effects of GH in peripheral tissues. Elevating GH through secretagogue stimulation rather than exogenous GH provides a more controlled research model because the feedback mechanisms remain intact and supraphysiological GH levels are less likely.
The original CJC-1295 RCT documented body composition changes alongside the hormonal measurements, with the sustained IGF-1 elevation providing the mechanistic basis for lean mass preservation effects studied in subsequent protocols. This area represents one of the most active ongoing research domains for both individual compounds and their combination.
Recovery and Repair
GH has well-established roles in tissue repair, collagen synthesis, and recovery from injury or surgery. IGF-1, which rises secondary to GH elevation, has direct effects on satellite cell activation in skeletal muscle, making it relevant to research on muscle recovery following damage or exertion. The combination of CJC-1295 and ipamorelin is studied in this context as a way to amplify the body's own repair machinery rather than supplying exogenous growth factors directly.
For researchers interested in how peptides approach recovery more broadly, the IGF-1 LR3 guide covers the downstream signaling cascade that GH secretagogues ultimately feed into, providing useful context on how elevated IGF-1 translates to tissue effects.
Age-Related GH Decline Research
GH secretion declines with age beginning in the third decade of life. By age 60, most individuals produce significantly less GH than they did at 20. This decline correlates with body composition changes, more fat, less muscle, reduced physical capacity, and slower tissue repair. Secretagogue research investigates whether restoring GH axis activity toward younger levels reverses or slows these age-related changes.
The preserved pulsatility finding from the Ionescu 2006 study is particularly relevant here: because CJC-1295 raises GH while maintaining the pulsatile pattern, it does not create the flat GH exposure profile associated with exogenous GH therapy, which can cause insulin resistance and other side effects.
Metabolic Research
GH and IGF-1 have significant effects on insulin sensitivity, glucose metabolism, and lipid profiles. GH generally promotes lipolysis and can transiently reduce insulin sensitivity, while the downstream IGF-1 elevation tends to improve insulin sensitivity. The net metabolic effect of sustained secretagogue stimulation depends on dose, timing, and individual metabolic baseline, making this an active research area with significant nuance to explore.
For context on how metabolic peptides broadly interact with fat storage, energy utilization, and body composition, the Peptides for Fat Loss guide provides a useful overview of the wider landscape.
CJC-1295 vs. Ipamorelin: Direct Comparison Across Key Parameters
For researchers deciding how to design protocols, the following comparison covers the practical differences between these two compounds across the dimensions that matter most:
- Receptor target: CJC-1295 targets the GHRH receptor on somatotrophs via cAMP/PKA signaling. Ipamorelin targets the GHS-R1a receptor via PLC/calcium signaling. These are independent pathways.
- Half-life: CJC-1295 with DAC has a half-life of 5.8 to 8.1 days due to albumin binding. CJC-1295 without DAC has a half-life of approximately 30 minutes. Ipamorelin has a half-life of approximately 2 hours.
- GH pulse profile: CJC-1295 with DAC elevates trough GH and mean GH over days. Ipamorelin produces acute GH peaks within 30 to 45 minutes of administration that return to baseline within 2 to 3 hours.
- IGF-1 elevation: CJC-1295 with DAC produces sustained IGF-1 elevation lasting 9 to 11 days from a single injection, rising 1.5 to 3-fold above baseline. Ipamorelin contributes to IGF-1 elevation but with a shorter time course.
- Selectivity: Ipamorelin is highly selective for GH release and does not significantly raise cortisol, ACTH, or prolactin. CJC-1295 acts through GHRH receptors which are relatively GH-selective, though cortisol effects have been less systematically studied.
- Hunger stimulation: Ipamorelin does not produce significant appetite stimulation. GHRP-6, a related but less selective GHRP, produces strong hunger effects. This makes ipamorelin preferable in research protocols where appetite effects would confound results.
- Administration frequency: CJC-1295 with DAC can be administered once weekly. Ipamorelin is typically administered daily or multiple times daily in research protocols to deliver multiple GH pulses.
Neither compound is categorically better than the other. They are better understood as having complementary profiles that researchers can use to answer different questions, or to combine for maximally amplified GH axis stimulation.
Research Considerations for CJC-1295 and Ipamorelin
Both CJC-1295 and ipamorelin are research compounds with no FDA approval for human therapeutic use. All published clinical data comes from trials conducted under formal research protocols with institutional oversight. Any use of these compounds outside properly authorized research settings is outside the scope of current regulatory standards.
Comparison to Tesamorelin
Tesamorelin is an FDA-approved GHRH analog and the closest approved comparator to CJC-1295. It is approved specifically for HIV-associated lipodystrophy. Comparing the research profiles of CJC-1295 and tesamorelin helps frame what regulatory approval of a GHRH analog requires and where CJC-1295 sits on that developmental pathway. The Tesamorelin guide covers the approved compound's evidence base and mechanism in detail.
Sourcing and Quality
Research-grade CJC-1295 and ipamorelin should both come with a Certificate of Analysis confirming identity by mass spectrometry, purity above 98%, and endotoxin testing. Peptide purity is particularly important for compounds studied in human-adjacent research contexts. The guide to How to Read a Certificate of Analysis explains what to look for when evaluating documentation from any peptide vendor.
Research-grade CJC-1295 and ipamorelin are available from vendors listed through PeptidesFinder. We may earn a commission from purchases made through links on this site, and we feature vendors whose quality standards meet the documentation requirements for legitimate preclinical research.
For converting mg/kg dosing to injection volumes based on solution concentration, a peptide calculator is an essential tool for any growth hormone secretagogue research protocol.
Frequently Asked Questions
What is the difference between a GHRH analog and a GHRP?
GHRH analogs like CJC-1295 mimic growth hormone-releasing hormone, the hypothalamic peptide that activates the GHRH receptor on pituitary somatotrophs. This stimulates GH synthesis and release through the cAMP/PKA second messenger pathway. GHRPs like ipamorelin mimic ghrelin, a stomach-derived peptide hormone, and activate the GHS-R1a receptor on the same cells through a separate PLC/calcium pathway. Both approaches increase GH output but through mechanistically independent systems, which is why combining them produces synergistic rather than merely additive effects.
Why is ipamorelin considered superior to GHRP-6?
Ipamorelin and GHRP-6 produce similar GH-releasing potency, but ipamorelin does not significantly elevate ACTH, cortisol, or prolactin, while GHRP-6 raises both ACTH and cortisol at GH-releasing doses. GHRP-6 also stimulates appetite more strongly due to broader hypothalamic activation. For research protocols where these secondary hormonal effects would confound results or create unwanted physiological changes, ipamorelin provides a cleaner research tool. For this reason it has become the preferred GHRP for most research applications where the goal is specifically GH axis stimulation without stress hormone activation.
What does DAC mean in CJC-1295 with DAC?
DAC stands for Drug Affinity Complex. It is a chemical modification that enables CJC-1295 to covalently bind to serum albumin after injection. Albumin is the most abundant protein in blood plasma and has a natural circulating half-life of approximately 19 days. By binding to albumin, CJC-1295 is protected from enzymatic degradation and cleared much more slowly than unmodified peptides. This is what gives CJC-1295 with DAC its 5.8 to 8.1 day half-life, transforming what would otherwise be a short-acting GHRH fragment into a once-weekly injectable compound capable of sustaining GH and IGF-1 elevation across the week.
Is the synergy between CJC-1295 and ipamorelin proven in humans?
The synergistic relationship between GHRH receptor activation and GHS-R1a receptor activation has been documented in human subjects in the form of GHRH plus GHRP combination studies. The foundational research confirmed that combined GHRH and GHRP administration produces GH release greater than the sum of either compound's individual response in normal men. The specific combination of CJC-1295 and ipamorelin exploits this same dual-receptor synergy, and while the majority of studies have tested the compounds individually rather than in formal combined trials, the mechanistic basis for their synergy is well-established in human physiology.
Can CJC-1295 and ipamorelin desensitize GH receptors over time?
Receptor desensitization is a genuine consideration with GH secretagogues. Continuous stimulation of any receptor system tends to downregulate receptor expression over time. The pulsatile dosing approach used with ipamorelin (typically with off periods between doses) is partly designed to minimize this. CJC-1295 without DAC, because it produces sharper peaks with recovery periods, carries less desensitization risk than constant-infusion GHRH. CJC-1295 with DAC maintains a chronic baseline elevation that theoretically could reduce GHRH receptor sensitivity over time with very prolonged use, though the preserved pulsatility observed in research suggests the system retains responsiveness under studied protocols. Long-term desensitization data in humans remains limited.
How do CJC-1295 and ipamorelin compare to exogenous HGH?
Exogenous human growth hormone (HGH) bypasses the pituitary entirely, delivering GH directly into the bloodstream in a single large dose. This produces a non-pulsatile GH exposure pattern that differs fundamentally from natural GH secretion and can suppress the pituitary's own GH production through negative feedback. CJC-1295 and ipamorelin work upstream by stimulating the pituitary to produce and release its own GH in a pattern that preserves pulsatility and natural feedback mechanisms. This means GH output through secretagogue stimulation cannot exceed what the pituitary is capable of producing, creating a physiological ceiling that exogenous HGH does not have. The clinical significance of this difference for long-term safety and efficacy remains an active research question.
Are CJC-1295 and ipamorelin on any prohibited substance lists?
Yes. Both CJC-1295 and ipamorelin are included on the World Anti-Doping Agency (WADA) prohibited list under the category of peptide hormones, growth factors, related substances, and mimetics. Both are detectable in urine and blood samples by modern mass spectrometry methods. This applies in all competitive sports governed by WADA rules. Additionally, both compounds are classified as research chemicals and are not approved for human therapeutic use by the FDA. Researchers working with these compounds do so under institutional authorization in properly controlled preclinical settings.
Conclusion
The comparison of CJC-1295 versus ipamorelin ultimately resolves to this: they are two complementary tools that target the same physiological goal through completely different molecular entry points. CJC-1295 acts as a sustained GHRH signal, extending the half-life of GHRH stimulation from minutes to days and maintaining elevated trough GH and IGF-1 between pulses. Ipamorelin delivers clean, selective GH pulses through the ghrelin receptor pathway without the cortisol, prolactin, or appetite confounders that limit older GHRPs.
The clinical evidence for CJC-1295 specifically is unusually strong for a research peptide, with randomized controlled trial data in human adults confirming dose-dependent GH and IGF-1 elevations, a well-characterized half-life, and an absence of serious adverse events. Ipamorelin's selectivity profile, established in rigorous head-to-head pharmacological testing against GHRP-2 and GHRP-6, makes it the cleanest tool available for GHS-R1a stimulation in research contexts.
Together, by targeting both axes of GH regulation simultaneously, they produce the kind of synergistic, physiologically patterned GH output that makes them the most commonly studied secretagogue pairing in the field. For researchers working in GH biology, aging, body composition, or recovery, understanding both compounds in depth is not optional. They are two sides of the same biological coin.
For a closer look at the FDA-approved GHRH analog that sits in the same mechanistic family as CJC-1295, the Tesamorelin guide provides important context on what the regulatory development of a GHRH analog looks like. And for the downstream signaling pathway that both CJC-1295 and ipamorelin ultimately feed into, the IGF-1 LR3 guide covers how elevated IGF-1 translates to tissue-level effects.
Watch for the upcoming Sermorelin Benefits: How It Restores HGH Levels Naturally, which examines another GHRH-pathway secretagogue with a long clinical history and a different pharmacokinetic profile from CJC-1295.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. CJC-1295 and ipamorelin are sold as research chemicals and are not FDA-approved for human use. Research peptides are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any research compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.