PT-141 (Bremelanotide): The Peptide for Libido and Sexual Health
PT-141 (bremelanotide) the only FDA-approved peptide that targets sexual desire through the brain. Learn how it works, clinical evidence, dosing, & safety 2026.
Most treatments for sexual dysfunction start in the wrong place.
They focus on the body. They increase blood flow, relax smooth muscle, and improve physical mechanics. These approaches can help with the physical side of sexual function, but they do very little for the most important factor of all: the brain's desire to engage in the first place.
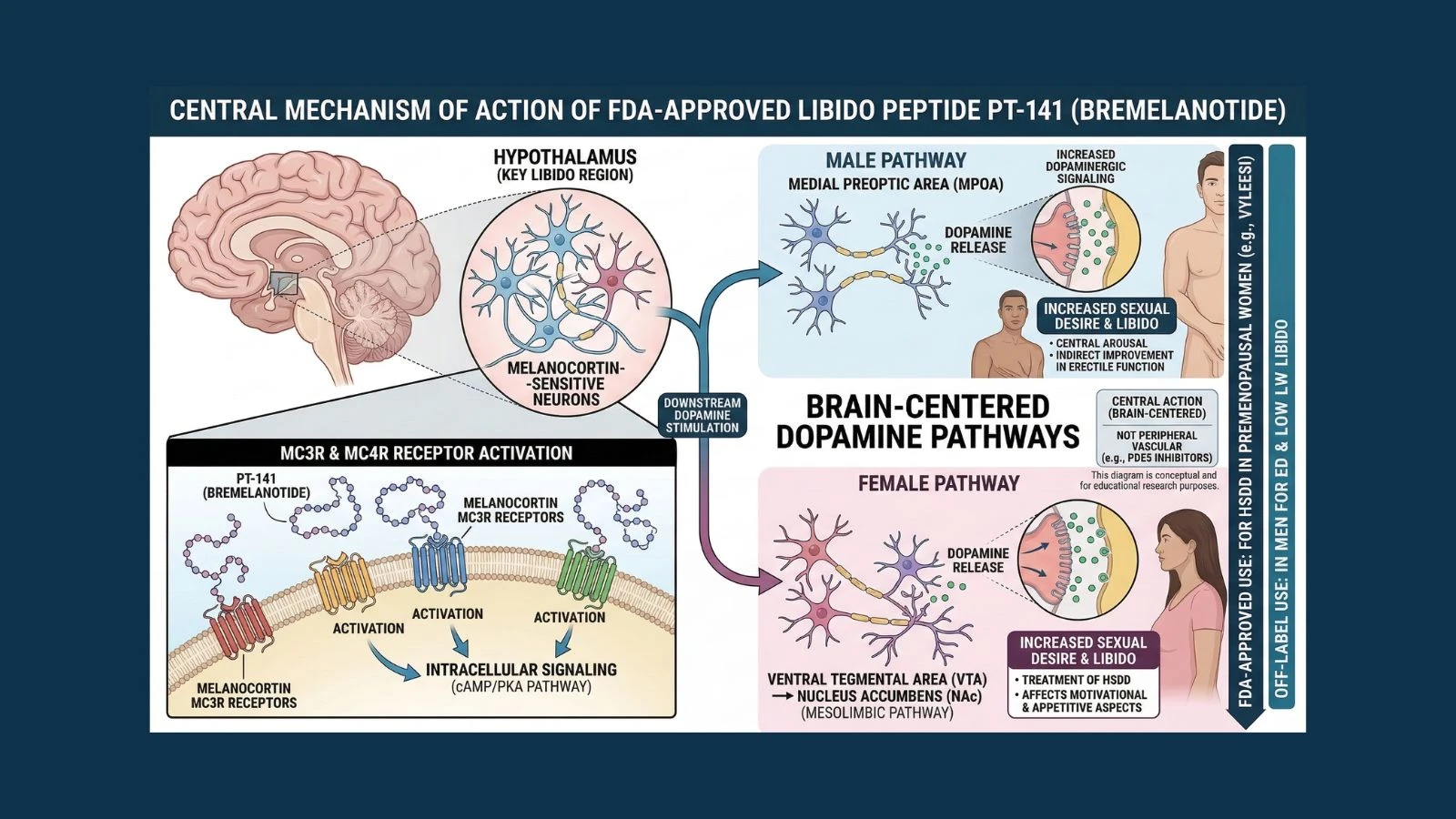
PT-141, also known as bremelanotide, is different. It is the first and only compound approved to treat sexual dysfunction that works through the brain rather than the body. Instead of targeting blood vessels or hormones, PT-141 activates a specific set of receptors in the hypothalamus that are directly connected to sexual motivation and arousal. It essentially tells the brain to want sex, rather than just making the body more capable of it.
For researchers and clinicians studying sexual health, low libido, hypoactive sexual desire disorder (HSDD), and the neuroscience of arousal, PT-141 represents a genuinely new class of treatment built on decades of melanocortin research. This guide covers exactly how it works, what the FDA approval process revealed, what the research shows for both men and women, and how to understand its place in the broader landscape of sexual health science.
New to the world of research peptides? Our Complete Beginner's Guide to Peptide Therapy gives you the foundational science you need before exploring any specific research compound.
What Is PT-141? From Sunless Tanning to Sexual Health
PT-141 has an unusual origin story. It was not designed to treat sexual dysfunction. It was born out of research into a sunless tanning compound.
In the 1980s, researchers at the University of Arizona were studying a peptide called alpha-MSH (alpha-melanocyte-stimulating hormone). Alpha-MSH activates melanocortin receptors on skin cells, stimulating melanin production and causing tanning. The team wanted to create a synthetic version that could tan skin without UV exposure.
They developed a compound called Melanotan I, and later a modified version called Melanotan II. During early clinical trials with Melanotan II, researchers noticed a consistent and unexpected side effect: strong spontaneous sexual arousal in both male and female participants. This was not the goal, but it was impossible to ignore.
Scientists realized that melanocortin receptors were doing far more than controlling skin pigmentation. Certain subtypes of these receptors, specifically MC3R and MC4R in the brain, were directly connected to sexual motivation pathways. Melanotan II was activating all melanocortin receptors at once, which also caused significant side effects including nausea and blood pressure changes.
PT-141 (bremelanotide) was developed as a refined version that targeted the sexual arousal pathway more selectively while reducing the broader melanocortin activation. It was stripped of the tanning effect and optimized for the brain-based arousal mechanism.
For context on the Melanotan compounds that preceded PT-141 and why their broader receptor activation created serious safety concerns, see our guide on Melanotan 2: The Science, Risks, and Reality.
FDA Approval: A Milestone in Sexual Health Medicine
In June 2019, the FDA approved bremelanotide under the brand name Vyleesi for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women. This made PT-141 the second FDA-approved treatment for female sexual dysfunction, alongside flibanserin (Addyi), but with a completely different mechanism of action.
The FDA approval was based on two Phase 3 randomized controlled trials reviewed by the agency, which demonstrated statistically significant improvements in sexual desire and reductions in distress related to low libido compared to placebo. Full prescribing information is available through the FDA drug label.
For men, PT-141 remains in the research and off-label clinical use category. Multiple studies have demonstrated significant efficacy for erectile dysfunction and low libido in men, including in cases where PDE5 inhibitors like sildenafil (Viagra) had failed, but a formal FDA approval for male sexual dysfunction has not yet been pursued through a complete approval pathway.
How PT-141 Works: The Brain-Centered Mechanism
Understanding how PT-141 works requires a brief look at the melanocortin system, one of the most complex and wide-ranging signaling networks in the human body.
The Melanocortin System
The melanocortin system consists of five receptor subtypes (MC1R through MC5R) distributed throughout the skin, brain, immune system, and other tissues. Each receptor subtype controls different biological functions:
- MC1R: Skin pigmentation and UV response
- MC2R: Stress hormone (cortisol) production in the adrenal glands
- MC3R: Energy balance, feeding behavior, and sexual function
- MC4R: Appetite regulation, energy expenditure, and sexual function
- MC5R: Exocrine gland secretion
PT-141 primarily targets MC3R and MC4R in the brain, particularly in the hypothalamus and limbic system. These are the receptors that govern the neural circuits associated with sexual motivation, arousal, and desire.
The Dopamine Connection
When PT-141 activates MC3R and MC4R in the hypothalamus, it triggers a downstream release of dopamine in the mesolimbic pathway, the brain's primary reward and motivation circuit. Dopamine is the neurotransmitter most associated with desire, anticipation, and motivation. It is what makes something feel worth pursuing.
By increasing dopamine activity in the circuits associated with sexual behavior, PT-141 creates a genuine neurological sense of sexual desire. This is not a peripheral effect. It is not improving blood flow or relaxing smooth muscle. It is directly modifying the brain's motivational state toward sexual interest.
Research published in PMC (PubMed Central) confirmed that melanocortin receptor activation in the paraventricular nucleus of the hypothalamus directly stimulates sexual behavior through oxytocin and dopamine pathways, establishing the neurological foundation for PT-141's mechanism of action.
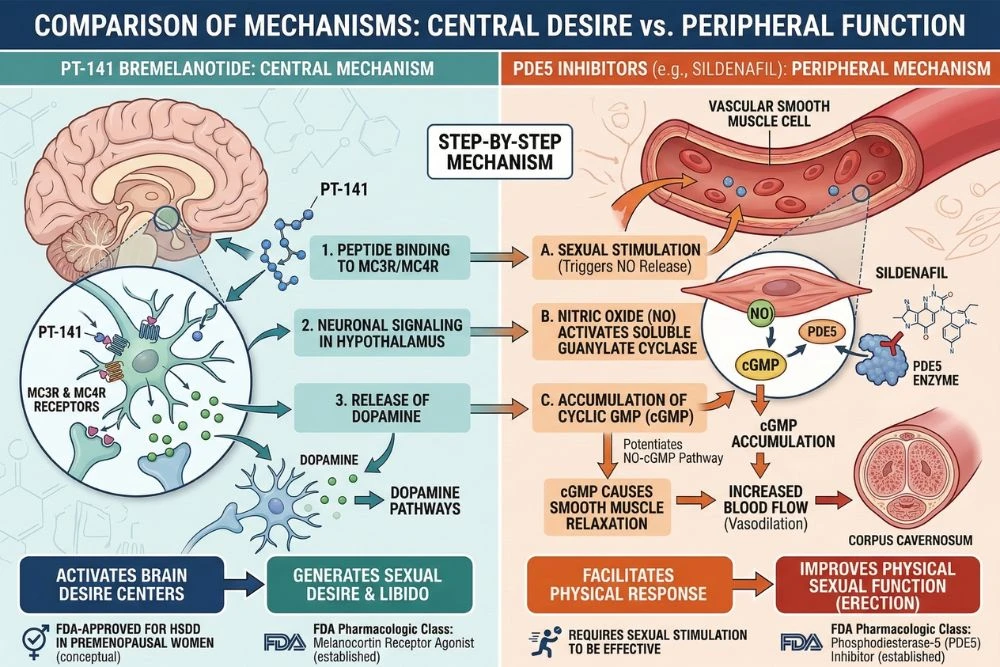
How PT-141 Differs from PDE5 Inhibitors
This distinction is critical for understanding PT-141's unique value. Drugs like sildenafil (Viagra) and tadalafil (Cialis) are PDE5 inhibitors. They work by relaxing the smooth muscle in blood vessels, increasing blood flow to genital tissue. They improve the body's physical capacity for sexual response but do nothing for the brain's desire to engage.
If someone has low libido, meaning the brain's motivation for sex is diminished, PDE5 inhibitors cannot address that. PT-141 works upstream of the physical response. It generates the desire first, which then leads to the physiological response naturally.
This is why PT-141 has shown efficacy in men with erectile dysfunction who had failed to respond to PDE5 inhibitors. Their physical equipment was not the limiting factor. The brain signal was. PT-141 addresses that brain signal directly.
PT-141 Research in Women: HSDD and Female Sexual Dysfunction
The majority of PT-141's clinical trial data comes from research in women, specifically those diagnosed with hypoactive sexual desire disorder (HSDD). HSDD is characterized by persistently low sexual desire that causes personal distress, and it is one of the most common forms of female sexual dysfunction, affecting an estimated 8 to 14 percent of women in the United States.
The Phase 3 Clinical Trials
The FDA approval of Vyleesi was supported by two large Phase 3 randomized controlled trials known as the RECONNECT studies. These trials enrolled premenopausal women diagnosed with HSDD and measured two primary outcomes: the number of satisfying sexual events per month and the level of distress associated with low sexual desire.
Results published in PMC Obstetrics and Gynecology showed that women using bremelanotide reported statistically significant improvements in both satisfying sexual events and desire scores compared to placebo, with the most meaningful improvements seen in women with more severe baseline HSDD symptoms.
Importantly, the improvement was in desire and motivation, not just physical response. Participants reported wanting sex more, not just being more physically responsive to it. This is the central distinction that makes PT-141 clinically meaningful for HSDD specifically.
How PT-141 Is Used for HSDD
The FDA-approved protocol for Vyleesi is a 1.75 mg subcutaneous injection administered approximately 45 minutes before anticipated sexual activity. It is not a daily medication. It is used on-demand, similar to how PDE5 inhibitors are used in men. The maximum recommended frequency is one dose per 24 hours and no more than one dose per day as needed.
The on-demand nature of PT-141 is one of its clinical advantages over daily oral medications like flibanserin (Addyi), which requires daily administration and has significant food, alcohol, and drug interaction considerations. PT-141 is taken only when desired and acts within a relatively predictable timeframe.
PT-141 vs. Flibanserin (Addyi) for Female Sexual Dysfunction
Both PT-141 and flibanserin are FDA-approved for HSDD in premenopausal women, but they work through completely different mechanisms. Flibanserin is a serotonin receptor partial agonist that modulates multiple neurotransmitter systems and must be taken daily. It has significant drug and alcohol interactions. PT-141 is a melanocortin receptor agonist taken on-demand with a more straightforward mechanism and fewer interaction concerns.
Clinical preference between the two depends on individual patient factors, but PT-141's on-demand dosing and direct melanocortin mechanism make it the more targeted and flexible option for many patients and researchers studying the neuroscience of female sexual desire.
PT-141 Research in Men: Libido, Erectile Function, and Beyond
While PT-141's FDA approval is specific to female HSDD, its research in men is extensive and the results are compelling. The same melanocortin pathway that drives female sexual desire operates in men, and PT-141 activates it equally effectively.
Erectile Dysfunction Research
Several published studies have examined PT-141's effect on erectile function in men with organic and psychogenic erectile dysfunction. The results have been consistently positive, with PT-141 producing measurable improvements in erectile response at doses ranging from 0.5 mg to 10 mg in published trial data.
A double-blind placebo-controlled study published in PubMed found that PT-141 produced clinically meaningful erectile responses in men with mild to moderate erectile dysfunction, including in a subset of men who had not responded adequately to sildenafil, confirming its value as an alternative mechanism for cases where standard PDE5 inhibitor therapy is insufficient.
Low Libido in Men
Beyond erectile function, PT-141 research has focused on male hypoactive sexual desire, which is less frequently discussed than female HSDD but affects a significant portion of men, particularly as testosterone declines with age.
Because PT-141 acts through dopamine pathways in the hypothalamus rather than through testosterone or sex hormones, it can address low libido even in men whose hormone levels are within normal range. This makes it particularly relevant for research into psychogenic low desire, where the issue is neurological rather than hormonal.
PT-141 as a Complement to Hormonal Therapies
In clinical and research contexts, PT-141 is sometimes combined with hormonal optimization protocols. Testosterone therapy can restore baseline sexual function and energy, but some men find their libido remains lower than expected even after testosterone levels are normalized. In these cases, PT-141's central mechanism can address the neurological component of desire that testosterone alone does not fully restore.
For researchers studying hormonal pathways and their relationship to sexual health, our guide on Kisspeptin: Restoring Testosterone and Hormonal Balance provides essential context on how the HPG axis governs testosterone production and its downstream effects on desire and motivation.

Dosing Protocols and Administration
PT-141 protocols vary between the FDA-approved clinical use and broader research applications. Here is what the published data shows for both contexts.
FDA-Approved Protocol (Vyleesi)
The approved protocol for bremelanotide (Vyleesi) in premenopausal women with HSDD is:
- Dose: 1.75 mg subcutaneous injection
- Timing: 45 minutes before anticipated sexual activity
- Frequency: As needed, maximum once per 24 hours
- Administration site: Abdomen or thigh via autoinjector
The 45-minute window allows time for the peptide to cross the blood-brain barrier, activate melanocortin receptors, and trigger the downstream dopamine response that generates the increase in sexual desire.
Research Dosing Ranges
In published research studies, PT-141 has been studied across a broader dose range depending on the study objective:
- Low range: 0.5 mg to 1 mg, used in dose-finding studies and sensitivity assessments
- Standard range: 1.25 mg to 1.75 mg, corresponding to the FDA-approved clinical dose and most commonly studied in human trials
- Higher range: Up to 10 mg, studied in early clinical trials for erectile dysfunction, though higher doses are associated with more pronounced side effects
Most current research and clinical use centers around the 1 to 1.75 mg range, which balances efficacy with tolerability.
Timing and Onset
PT-141's onset of action is typically 30 to 60 minutes after subcutaneous injection. Peak effects are generally reported between 1 and 3 hours post-injection. The duration of enhanced sexual desire and arousal typically lasts 6 to 12 hours, though individual variation is significant.
Unlike PDE5 inhibitors, which require sexual stimulation to produce their effects, PT-141's central mechanism means that enhanced desire can occur spontaneously, without requiring external arousal stimuli. This is one of its defining characteristics and a key part of its therapeutic distinction.
Reconstitution for Research Use
For research applications, PT-141 is typically supplied as lyophilized powder and reconstituted with bacteriostatic water. Reconstituted solutions should be stored refrigerated and protected from light.
For accurate peptide reconstitution and dosing calculations, our Peptide Calculator Guide walks through the complete process.
Safety Profile: What the Clinical Trials Revealed
PT-141's safety profile is one of the most thoroughly documented of any research peptide because of the extensive clinical trial program required for FDA approval. Here is what that data shows.
Most Common Side Effects
In the Phase 3 RECONNECT trials, the most commonly reported side effects were:
- Nausea: The most frequently reported side effect, occurring in approximately 40 percent of participants. Nausea typically begins within an hour of injection and resolves within a few hours. In clinical practice, taking PT-141 with a light meal and remaining in an upright position can reduce nausea severity
- Flushing: Reported in approximately 20 percent of participants, typically presenting as facial warmth and redness related to transient blood pressure changes
- Injection site reactions: Mild localized redness, bruising, or discomfort at the subcutaneous injection site, reported in approximately 13 percent of participants
- Headache: Reported in approximately 11 percent of participants, generally mild and self-resolving
Blood Pressure Considerations
PT-141 can cause a transient decrease in blood pressure followed by a transient increase. In the clinical trials, the mean maximum decrease in systolic blood pressure was approximately 6 mmHg and occurred within 12 hours of dosing. For most healthy participants this is not clinically significant, but it is an important consideration for research subjects with cardiovascular conditions or those taking antihypertensive medications.
The prescribing information recommends against use in individuals with known cardiovascular disease. Full prescribing details and contraindications are outlined in the official FDA label for Vyleesi.
Hyperpigmentation with Frequent Use
Because PT-141 is a melanocortin agonist, it retains some degree of melanocyte-stimulating activity even though it is far more selective than Melanotan II. With frequent or high-dose use over extended periods, some individuals have reported focal hyperpigmentation, particularly on the face, breasts, and gums. The FDA label notes this risk and recommends against use in individuals with dark skin tones who may be more susceptible to this effect.
At the approved dose and on-demand frequency, this risk appears to be minimal in clinical practice, but it is an important monitoring consideration in research protocols involving frequent administration.
Contraindications
PT-141 (Vyleesi) is contraindicated in individuals with known cardiovascular disease, uncontrolled hypertension, or high risk of cardiovascular events. It should not be used in postmenopausal women or men as an approved medication (though research continues in these populations). It is also not indicated for use in combination with certain antihypertensive medications due to the risk of additive blood pressure lowering effects.
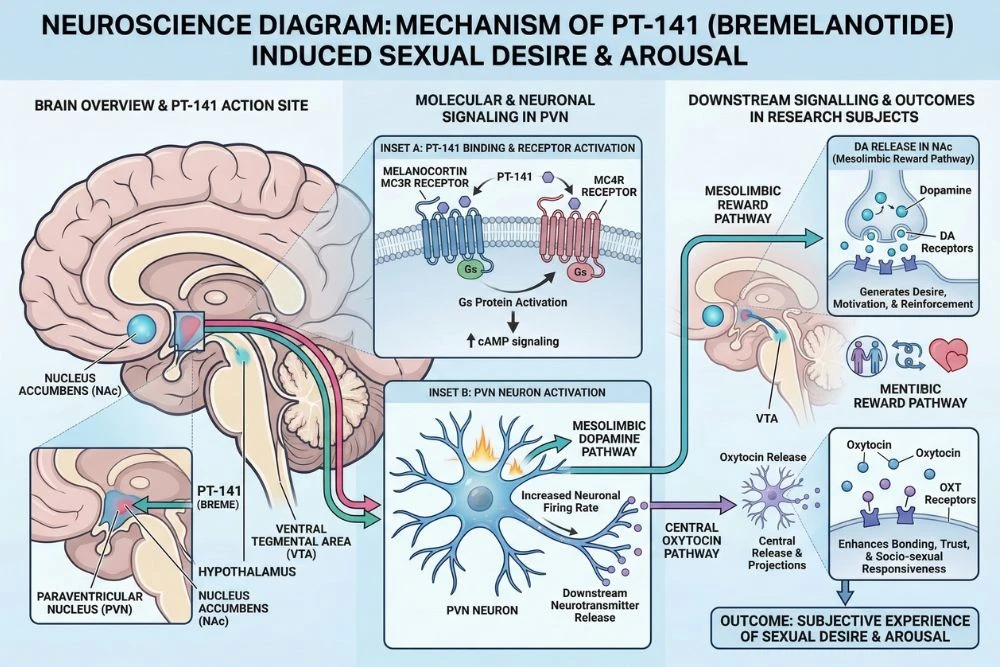
The Neuroscience of Sexual Desire: Why the Brain Matters
PT-141's mechanism gives researchers a unique window into how sexual desire is generated and regulated in the brain. This section explores what that research has revealed about the neuroscience of arousal.
Sexual Desire as a Brain-Generated State
Modern neuroscience has established that sexual desire is not simply a hormonal phenomenon or a genital response. It is a specific motivational state generated by neural circuits in the hypothalamus, limbic system, and prefrontal cortex. These circuits integrate hormonal signals, sensory inputs, memories, emotional state, and stress levels to produce the subjective experience of wanting sex.
The melanocortin system is one of the key molecular regulators of these circuits. When MC3R and MC4R in the hypothalamus are activated, they trigger a cascade that includes dopamine release, oxytocin release, and modulation of the paraventricular nucleus, which governs the physiological preparation for sexual activity.
Why Stress and Anxiety Kill Libido
The melanocortin system also explains why stress and anxiety so reliably suppress sexual desire. Cortisol and other stress hormones antagonize MC3R and MC4R signaling in the hypothalamus, effectively blocking the neural pathway that generates sexual motivation. When someone is chronically stressed, the brain's melanocortin system is partially suppressed, and libido drops as a result.
PT-141 activates these receptors directly, partially bypassing the cortisol suppression. This is why research has shown PT-141 can be effective even in individuals whose low libido is primarily psychogenic or stress-related rather than hormonal.
The Role of Oxytocin
Beyond dopamine, PT-141's melanocortin activation also stimulates oxytocin release in the paraventricular nucleus. Oxytocin, often called the bonding or connection hormone, plays a key role in sexual arousal, orgasm, and the emotional connection associated with intimacy. PT-141's stimulation of the oxytocin pathway is thought to contribute to the increased sense of emotional connection and intensity of arousal that some research participants have reported alongside the increase in desire.
For a broader look at how neuropeptides govern brain states including arousal and anxiety, our guide on Neuropeptide S: The Science of Arousal, Anxiety, and Neurological Research explores the intersection of peptide science and brain function.
PT-141 vs. Other Approaches to Sexual Health
Understanding where PT-141 fits in the broader landscape of sexual health treatments helps researchers and clinicians make more informed decisions about which approach is appropriate for different research questions and patient profiles.
PT-141 vs. Testosterone Therapy
Testosterone is the primary hormonal driver of sexual desire in both men and women. When testosterone is genuinely deficient, restoring it is the most direct approach to improving libido. But testosterone therapy does not help everyone with low libido because the issue is not always hormonal. PT-141 addresses the neural desire pathway directly, making it useful in cases where testosterone is normal but desire is still low. The two approaches are complementary rather than competing.
PT-141 vs. Sildenafil and Tadalafil
PDE5 inhibitors address physical sexual function. PT-141 addresses psychological sexual desire. In men with both low libido and erectile dysfunction, combining the two approaches targets different parts of the problem simultaneously. PT-141 generates the brain's desire to engage, while the PDE5 inhibitor ensures the physical response follows reliably. This combination has been studied in research settings and is increasingly used in clinical practice for complex cases.
PT-141 vs. Flibanserin
Both are FDA-approved for female HSDD. Flibanserin requires daily dosing, has significant drug and alcohol interactions, and works through a more complex multi-receptor mechanism. PT-141 is on-demand, has a cleaner pharmacological profile, and works through a well-defined melanocortin mechanism. For patients seeking flexibility and a more predictable dosing experience, PT-141 generally offers practical advantages. For patients who prefer not to use injectables, flibanserin's oral tablet form may be preferred despite its limitations.
Sourcing Quality PT-141 for Research
For research applications outside of the FDA-approved Vyleesi formulation, PT-141 is available as a research-grade peptide. Quality verification is essential, as with all research peptides.
What a Certificate of Analysis Should Confirm
Any PT-141 sourced for research should come with third-party laboratory documentation confirming:
- HPLC purity at or above 98 percent
- Mass spectrometry verification of the correct molecular weight (approximately 1025.2 Da for bremelanotide)
- Amino acid sequence confirmation
- Batch number and accredited third-party laboratory certification
For a step-by-step walkthrough of what to look for in a peptide CoA, our guide on How to Read a Peptide Certificate of Analysis covers every section in plain language.
Recommended Research Supplier
Peptides Finder connects researchers with vetted suppliers providing full third-party testing documentation for PT-141 and all major research peptides.
Browse PT-141 Research Compounds at Peptides Finder
We may earn a commission from purchases made through links on this page.
Frequently Asked Questions
What is PT-141 and how does it work?
PT-141, also known as bremelanotide, is a synthetic peptide that activates melanocortin receptors MC3R and MC4R in the hypothalamus. This activation triggers dopamine and oxytocin release in brain circuits associated with sexual motivation and desire. Unlike most sexual health treatments that work on blood flow or hormones, PT-141 works directly in the brain to generate the neurological state of sexual desire. It is the only compound that has been FDA-approved specifically because of this central, brain-based mechanism.
Is PT-141 FDA approved?
Yes. PT-141 received FDA approval in June 2019 under the brand name Vyleesi for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women. The approval was based on two Phase 3 randomized controlled trials that demonstrated statistically significant improvements in sexual desire and reductions in HSDD-related distress compared to placebo. For men and for postmenopausal women, PT-141 is used in clinical research and off-label contexts but does not currently hold a formal FDA approval for these populations.
How is PT-141 different from Viagra?
Viagra (sildenafil) and other PDE5 inhibitors work peripherally by relaxing blood vessel smooth muscle to increase blood flow to genital tissue. They improve the physical mechanics of sexual response but do not affect the brain's desire to engage. PT-141 works centrally in the brain, activating the neural circuits that generate sexual motivation and desire. This means PT-141 can be effective in cases where the issue is low libido rather than physical erectile or arousal dysfunction, and it has shown efficacy in men who did not respond adequately to PDE5 inhibitors.
What are the main side effects of PT-141?
The most common side effects observed in Phase 3 clinical trials were nausea (approximately 40 percent of participants), flushing (approximately 20 percent), injection site reactions (approximately 13 percent), and headache (approximately 11 percent). A transient blood pressure decrease can also occur. Nausea is the most significant tolerability concern and can be reduced by administering PT-141 with a light meal and remaining upright for an hour after injection. With frequent or high-dose use, focal hyperpigmentation is a possible long-term concern given PT-141's melanocortin activity.
How long does PT-141 take to work?
PT-141 typically begins to produce effects within 30 to 60 minutes of subcutaneous injection. Peak effects on sexual desire and arousal are generally reported between 1 and 3 hours post-injection. The duration of enhanced sexual motivation typically lasts 6 to 12 hours, though individual responses vary considerably. The approved protocol recommends administration approximately 45 minutes before anticipated sexual activity to allow time for the brain-based mechanism to activate.
Can PT-141 be used by both men and women?
Yes. The underlying melanocortin mechanism that PT-141 activates operates in both male and female brains. The FDA approval is specific to premenopausal women with HSDD, but clinical research has studied PT-141 in men with erectile dysfunction and low libido with consistently positive results. Multiple published studies have demonstrated significant improvements in erectile function and sexual desire in men, including in cases where PDE5 inhibitors were ineffective. Off-label clinical use in men is established and supported by the research literature.
Does PT-141 affect hormones?
PT-141 does not directly alter sex hormone levels such as testosterone or estrogen. Its mechanism is neurological rather than endocrine. It activates brain receptors to generate a desire state through dopamine and oxytocin pathways without changing the hormonal environment. This is one of its advantages over hormonal therapies: it can address low libido caused by psychological or neurological factors even when hormone levels are within normal range. However, because it stimulates oxytocin and has some broader melanocortin activity, it may interact with other hormonal systems in ways that ongoing research continues to explore.
Conclusion: PT-141 and the New Science of Sexual Desire
PT-141 represents a genuine shift in how science approaches sexual health. For decades, the field focused almost entirely on the peripheral, physical side of sexual function. Blood flow. Tissue mechanics. Hormones. PT-141 changed the conversation by demonstrating that the brain is the most important organ in human sexuality, and that directly targeting the brain's desire circuits is both possible and effective.
The approval of Vyleesi for female HSDD was more than a regulatory milestone. It was a scientific validation of the melanocortin pathway's central role in human sexual motivation. And it opened the door to a new generation of research into how desire, arousal, and sexual health are generated and regulated at the neural level.
For researchers studying sexual dysfunction, psychogenic low libido, the neuroscience of arousal, or the intersection of brain chemistry and intimate health, PT-141 is an essential compound to understand. Its well-documented mechanism, extensive clinical trial data, and approved status make it one of the most credible and thoroughly studied research peptides in the sexual health space.
Whether studied in isolation or as part of a comprehensive sexual health protocol combining hormonal optimization and central desire activation, PT-141 stands as a benchmark for brain-based approaches to libido restoration.
Explore the full library of sexual health, hormonal, and neurological peptide research guides at Peptides Finder, and stay current with the latest developments in peptide science.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. Bremelanotide (PT-141) is FDA-approved as Vyleesi for hypoactive sexual desire disorder in premenopausal women and is available by prescription only. Research-grade PT-141 is sold as a research chemical and is intended strictly for laboratory research, not for human consumption outside of licensed medical supervision. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.