Melanotan 2: The Science, Risks, and Reality Behind the 'Barbie Drug'
What is Melanotan 2? Understand the science, tanning effects, libido impact, and serious health risks including FDA warnings. Complete safety guide 2026.
CRITICAL SAFETY WARNING
Melanotan 2 is NOT approved by the FDA, TGA (Australia), or any major regulatory body for human use. It is illegal to sell or market for cosmetic tanning or sexual enhancement in the United States, United Kingdom, and Australia. This article is for educational and research purposes only. The health risks are serious and potentially life-threatening.
Social media calls it the "Barbie Drug."
TikTok influencers promise a year-round tan without sunlight. Underground websites market it as a miracle peptide for both bronzed skin and explosive libido. And thousands of people inject themselves with a substance that was abandoned by pharmaceutical companies decades ago due to safety concerns.
This is Melanotan 2, and its story is equal parts fascinating science and cautionary tale.
Originally developed in the 1980s at the University of Arizona as a potential skin cancer preventative, Melanotan 2 is a synthetic analog of alpha-melanocyte-stimulating hormone (α-MSH) that activates melanocortin receptors throughout the body. Clinical trials revealed it could indeed darken skin pigmentation. But researchers also discovered two unexpected effects: dramatic appetite suppression and spontaneous erections lasting hours.
These "side effects" made Melanotan 2 commercially appealing, but the safety profile made it commercially impossible. Development was halted. Patents were sold. And what was supposed to disappear into pharmaceutical history instead went underground, where it thrives today as an unregulated research chemical sold on the gray market.
This guide provides a complete scientific overview of Melanotan 2: its mechanism of action, the clinical evidence for its effects, and most importantly, the serious health risks that have prompted warnings from health authorities worldwide.
What Is Melanotan 2? A Synthetic Melanocortin Analog
To understand Melanotan 2, you need to understand the natural hormone it mimics.
Alpha-melanocyte-stimulating hormone (α-MSH) is a naturally occurring peptide derived from a larger precursor protein called proopiomelanocortin (POMC). POMC is processed in the pituitary gland and hypothalamus into several bioactive peptides, including α-MSH, β-MSH, and adrenocorticotropic hormone (ACTH).
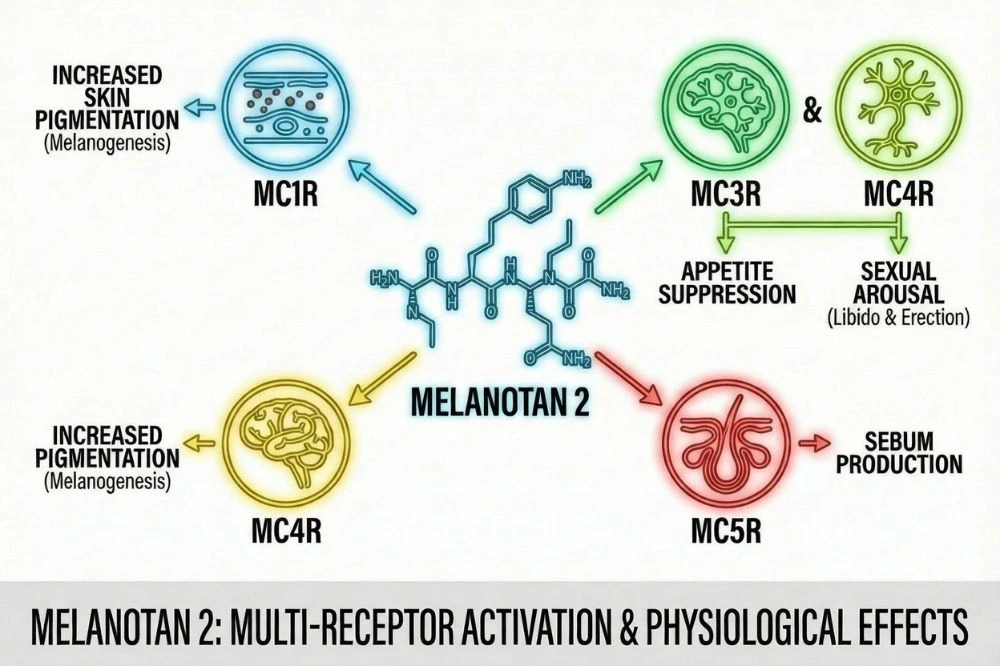
These melanocortin peptides bind to five different G-protein coupled receptors called melanocortin receptors (MC1R through MC5R), according to research published in PMC. Each receptor has distinct tissue distribution and physiological functions:
- MC1R: Found in skin melanocytes. Regulates pigmentation and tanning response.
- MC2R: Located in the adrenal gland. Mediates cortisol release in response to stress.
- MC3R: Expressed in the brain and periphery. Involved in energy homeostasis and feeding behavior.
- MC4R: Widely distributed in the central nervous system. Controls appetite, sexual function, energy expenditure, and cardiovascular function.
- MC5R: Found throughout the body. Regulates exocrine gland secretion.
Melanotan 2 is a cyclic heptapeptide (a ring-shaped chain of seven amino acids) that was designed to be a more potent and longer-lasting analog of α-MSH. Its full chemical sequence is: Ac-Nle⁴-c[Asp⁵-His⁶-D-Phe⁷-Arg⁸-Trp⁹-Lys¹⁰]-NH₂.
The key modification is the cyclization (forming a ring structure), which makes the peptide resistant to enzymatic degradation and dramatically extends its half-life compared to natural α-MSH. While α-MSH is broken down within minutes in the bloodstream, Melanotan 2 remains active for hours.
Unlike its predecessor Melanotan 1 (afamelanotide, which was FDA-approved in 2019 for a rare genetic condition called erythropoietic protoporphyria), Melanotan 2 is a nonselective melanocortin receptor agonist. This means it activates multiple melanocortin receptors, not just MC1R.
This lack of selectivity is both its strength and its danger.
The Mechanism: How Melanotan 2 Works on Multiple Systems
When injected subcutaneously or administered as a nasal spray, Melanotan 2 enters the bloodstream and crosses the blood-brain barrier. It then binds to melanocortin receptors in multiple tissues simultaneously.
Effect 1: Skin Pigmentation via MC1R
In the skin, Melanotan 2 binds to MC1R on melanocytes (pigment-producing cells). This triggers a signaling cascade that increases production of eumelanin, the brown-black pigment responsible for skin darkening.
According to the Australian Therapeutic Goods Administration (TGA), users can experience "dramatic skin darkening in just days" when combined with UV exposure. This rapid tanning effect is what drives demand for the product.
Critical point: Melanotan 2 does NOT provide sun protection. It stimulates melanin production but does not prevent UV damage to DNA. The tan it produces offers minimal (if any) protective benefit against skin cancer.
Effect 2: Appetite Suppression via MC4R
MC4R activation in the hypothalamus powerfully suppresses appetite and increases energy expenditure. Research in PubMed demonstrates that melanocortin receptor agonists like Melanotan 2 can reduce food intake and increase metabolic rate in animal models.
This effect explains the weight loss commonly reported by users. However, MC4R activation can also cause nausea, a side effect so common that it occurs in the majority of users. In clinical trials, 12.9% of subjects experienced severe nausea at standard doses.
Effect 3: Sexual Function via MC4R
This is where Melanotan 2 becomes particularly interesting from a neuroscience perspective.
MC4R is expressed throughout brain regions involved in sexual motivation, arousal, and genital response, including the hypothalamus, nucleus accumbens, and spinal cord. When Melanotan 2 activates these receptors, it triggers a central nervous system-mediated sexual response.
This is fundamentally different from drugs like sildenafil (Viagra), which work by increasing blood flow to the genitals. Melanotan 2 acts on brain circuits that control sexual desire and arousal.
Clinical studies published in the Journal of Urology found that:
- In men with erectile dysfunction, Melanotan 2 initiated erections in 17 of 20 subjects (85%) without sexual stimulation.
- Mean duration of rigidity greater than 80% was 41 minutes.
- 68% of doses were associated with increased sexual desire compared to 19% with placebo.
Research in both men and women showed enhanced sexual function, including increased genital arousal in females.
These sexual effects were so pronounced that Palatin Technologies licensed Melanotan 2 specifically to develop it as a treatment for sexual dysfunction. They eventually created a derivative called bremelanotide (PT-141), which was FDA-approved in 2019 for hypoactive sexual desire disorder in women under the brand name Vyleesi.
The difference? Bremelanotide underwent rigorous clinical trials, safety testing, and regulatory approval. Melanotan 2 did not.
Why Melanotan 2 Was Never Approved for Human Use
If Melanotan 2 works so well, why isn't it an FDA-approved medication?
The answer lies in its risk-to-benefit profile.
Development Was Abandoned Due to Safety Concerns
Melanotan 2 was originally developed by researchers at the University of Arizona in the late 1980s and early 1990s. The initial goal was to create a drug that would stimulate tanning without UV exposure, potentially reducing skin cancer rates.
However, as clinical development progressed, several issues emerged:
- Lack of receptor selectivity led to widespread systemic effects.
- High incidence of side effects (nausea, flushing, yawning, darkening of existing moles).
- Cardiovascular concerns including increased blood pressure and heart rate.
- Priapism risk (prolonged, painful erections requiring medical intervention).
- Unknown long-term effects on melanocyte behavior and melanoma risk.
Clinuvel Pharmaceuticals, which licensed Melanotan 2 for cosmetic development, abandoned the project in the 2000s due to regulatory restrictions and concerns about promoting sun-seeking behavior, according to Wikipedia.
FDA and International Warnings
The FDA has explicitly warned against Melanotan 2 use and issued warning letters to companies attempting to sell it. In a 2007 warning letter, the FDA stated that Melanotan 2 is "being marketed and sold illegally as a preventative against skin cancer and as a tanning agent," according to archived FDA communications.
Health authorities in Australia, the UK, and across Europe have issued similar warnings. The TGA explicitly states that Melanotan 2:
- Has NOT been assessed for quality or safety.
- Could contain toxic, poor quality, or counterfeit ingredients.
- Poses "serious side effects that can be very damaging to your health."
It is illegal to advertise or supply Melanotan 2 in Australia, and similar restrictions exist in most developed countries.
Serious Health Risks: What the Research Shows
The health risks of Melanotan 2 are not theoretical. They are documented in peer-reviewed medical literature.
Common Side Effects (Experienced by Most Users)
- Nausea and vomiting: Reported in the majority of users. Can be severe in 13% of cases.
- Facial flushing: Redness and warmth due to peripheral vasodilation.
- Yawning and stretching: Compulsive, repetitive yawning lasting hours.
- Loss of appetite: Significant reduction in food intake.
- Darkening of moles and freckles: Existing pigmented lesions become darker and more prominent.
- Spontaneous erections: In males, often occurring without sexual stimulation.
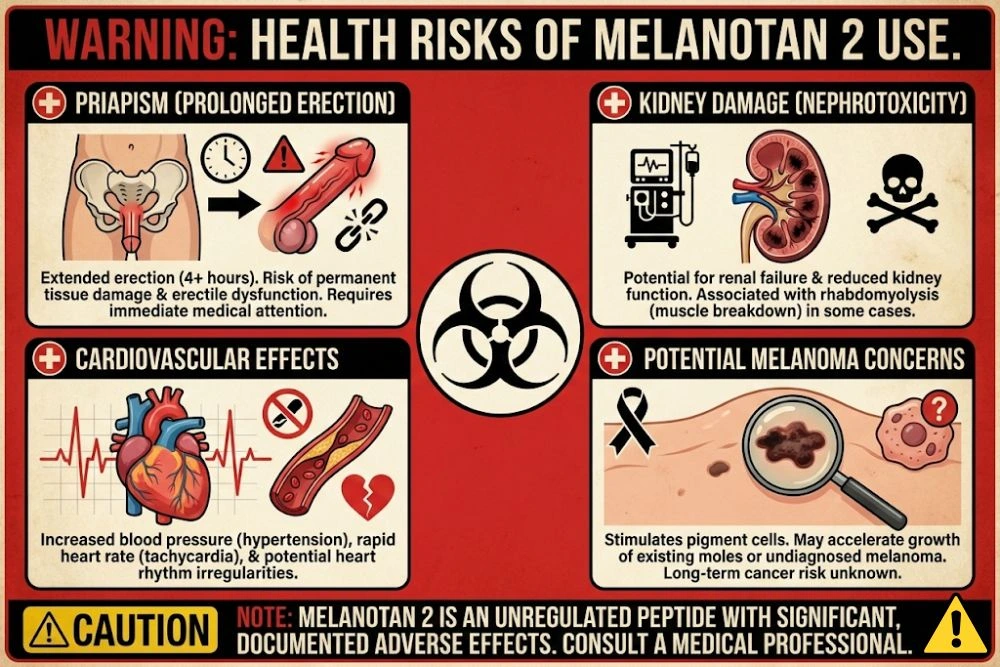
Serious Adverse Events (Documented in Case Reports)
According to Cleveland Clinic and other medical sources, Melanotan 2 has been associated with:
- Priapism (prolonged, painful erections lasting more than 4 hours). This is a medical emergency that can cause permanent erectile dysfunction and tissue damage if not treated immediately.
- Renal infarction (kidney damage due to reduced blood flow). Multiple case reports in medical literature document kidney failure following Melanotan 2 use.
- Rhabdomyolysis (breakdown of muscle tissue that releases proteins into the blood, damaging kidneys). This condition can be fatal.
- Hypertension (elevated blood pressure and heart rate from sympathetic nervous system activation).
- Brain swelling (cerebral edema), a rare but potentially fatal complication.
The Melanoma Question: Does Melanotan 2 Cause Skin Cancer?
This is the most controversial and scientifically uncertain aspect of Melanotan 2 safety.
There have been documented cases of melanoma appearing or worsening in Melanotan 2 users. However, establishing causation is difficult because:
- People who use Melanotan 2 often engage in sun-seeking behavior (tanning beds, unprotected sun exposure).
- Many users have fair skin (a melanoma risk factor) and want to tan without burning.
- Long-term controlled studies have never been conducted.
A 2021 scientific review concluded that "the increased risk of melanoma in Melanotan users, who use it for tanning and exhibit sun-seeking behaviour, can probably be explained by more UV exposure," according to research cited on Wikipedia.
However, the same review notes that Melanotan 2 causes rapid darkening and proliferation of moles, which is inherently concerning from a dermatological perspective. The TGA warns specifically about "increased moles and freckles" as a documented side effect.
Bottom line: The melanoma risk is unquantified but cannot be ruled out, especially with chronic use. Anyone using Melanotan 2 should undergo regular full-body skin checks by a dermatologist. To learn more about peptide safety and quality control, read our guide on Scientific Peptides: How to Read a Certificate of Analysis.
The Unregulated Market: Contamination and Quality Control Issues
Even if you accept the known risks of Melanotan 2, there's another layer of danger: you have no idea what's actually in the vial.
Because Melanotan 2 is not approved for human use, it's sold as a "research chemical" with the disclaimer "not for human consumption." There is no regulatory oversight, no quality control, and no accountability.
Studies analyzing Melanotan 2 products purchased online have found:
- Actual peptide content ranging from 4.3 mg to 8.8 mg in vials labeled as containing 10 mg.
- Up to 6% of product mass consisting of unknown impurities.
- Potential bacterial contamination from non-sterile manufacturing.
- Mislabeling and counterfeit products.
According to a study published in The Conversation, products marketed as Melanotan 2 "are not held to the high safety standards as TGA-approved products. This could result in variability in dose, undeclared ingredients and potential microbial contamination."
When you inject or inhale an unregulated substance, you're essentially participating in an uncontrolled human experiment.
Social Media, Influencers, and the "Barbie Drug" Phenomenon
Despite FDA warnings, TGA crackdowns, and documented health risks, Melanotan 2 continues to thrive on social media.
TikTok and Instagram influencers promote it as a safe way to achieve year-round tan skin. The hashtags #melanotan and #melanotan2 were banned on TikTok, but content continues to appear under generic tags like #tanning and #peptides.
Why is it so popular?
- Cultural beauty standards that prioritize tanned skin, especially in fitness and bodybuilding communities.
- The "Barbie" branding (tan skin, thin body, enhanced libido) appeals to people seeking the "perfect" physique.
- Misinformation from influencers who downplay or omit safety risks.
- Easy online access to underground suppliers.
According to a study on Melanotan 2 marketing, "social media has been driving unlicensed melanotan-II sales" with influencers providing endorsements and testimonials that fail to mention serious health consequences.
The Cancer Council in Australia has launched a campaign called "End the Trend" specifically targeting the normalization of tanning on social media.
Legitimate Research Applications: What Science Has Learned
While Melanotan 2 should not be used by the general public, it has contributed valuable insights to melanocortin receptor research.
Sexual Dysfunction Research
The sexual effects of Melanotan 2 led to the development of bremelanotide (PT-141), which received FDA approval for treating hypoactive sexual desire disorder in premenopausal women. This demonstrates that melanocortin-based therapies can be developed safely when subjected to proper clinical trials and regulatory oversight.
Obesity and Appetite Regulation
Melanotan 2 has been instrumental in understanding the role of MC4R in energy homeostasis. Research published in PMC shows that MC4R mutations are the most common monogenic cause of obesity in humans.
This research has led to development of selective MC4R agonists for obesity treatment, which are being evaluated in clinical trials.
Neuroprotection and Other Applications
Studies in animal models have shown that Melanotan 2 may have neuroprotective properties, promoting peripheral nerve regeneration after injury. However, these findings are preliminary and do not justify human use outside controlled research settings.

Safer Alternatives for Tanning and Sexual Enhancement
If you want tanned skin or improved sexual function, there are safer, legal, FDA-approved options.
For Tanning
- Topical self-tanners containing dihydroxyacetone (DHA) are FDA-approved and safe. They stain the outermost layer of skin without affecting melanocytes or requiring UV exposure.
- Professional spray tans offer consistent, controlled results without systemic absorption.
- Gradual tanning moisturizers provide natural-looking color buildup.
For Sexual Dysfunction
- PDE5 inhibitors (sildenafil, tadalafil) for erectile dysfunction in men.
- Bremelanotide (Vyleesi) for hypoactive sexual desire disorder in women (FDA-approved).
- Hormone therapy under medical supervision for hormone-related sexual dysfunction.
- Counseling and therapy for psychological factors.
Want to learn about FDA-approved peptide therapies for sexual health? See PT-141 (Bremelanotide): The Peptide for Libido and Sexual Health
Frequently Asked Questions
Is Melanotan 2 legal to buy?
No. Melanotan 2 is not approved by the FDA, TGA, or any major regulatory body for human use. It is illegal to sell or market it for cosmetic tanning or sexual enhancement in the United States, United Kingdom, Australia, and most of Europe. It may be sold as a "research chemical" with disclaimers, but this does not make it legal or safe for personal use.
What's the difference between Melanotan 1 and Melanotan 2?
Melanotan 1 (afamelanotide) is a linear peptide that primarily activates MC1R (skin pigmentation receptor). It was FDA-approved in 2019 under the brand name Scenesse for treating erythropoietic protoporphyria, a rare genetic condition. Melanotan 2 is a cyclic peptide that nonselectively activates MC1R, MC3R, MC4R, and MC5R, producing broader effects including appetite suppression and sexual arousal. Melanotan 2 is NOT approved for any medical use.
Can Melanotan 2 protect against skin cancer?
No. While Melanotan 2 stimulates melanin production, the resulting tan provides minimal (if any) protection against UV-induced DNA damage. The TGA explicitly states that "no tan, fake or real, will protect skin against damage from sun exposure." Using Melanotan 2 does not reduce your need for sunscreen or sun protection.
How long do the effects of Melanotan 2 last?
The tanning effect can persist for weeks to months after discontinuation, gradually fading as the skin naturally sheds melanin-rich cells. The sexual and appetite effects are acute, lasting hours after each injection. The darkening of moles and freckles may be permanent.
Are nasal spray versions safer than injections?
No. Nasal sprays may actually be more dangerous because the mucous membranes in the nose provide direct access to the bloodstream, allowing rapid systemic absorption. According to Cleveland Clinic, "anything that goes into your nose will be absorbed into your bloodstream much more quickly" than through skin. The risks are the same regardless of route of administration.
What should I do if I've already used Melanotan 2?
Stop using it immediately and consult a healthcare provider. Schedule a full-body skin examination with a dermatologist to check for any concerning changes in moles or new lesions. If you experience symptoms like decreased urination, severe muscle pain, chest pain, or prolonged erection, seek emergency medical care and disclose your Melanotan 2 use.
Conclusion: The Risk Is Not Worth the Reward
Melanotan 2 represents a fascinating chapter in melanocortin receptor research. Its ability to activate multiple physiological systems through a single peptide has taught us valuable lessons about neurobiology, appetite regulation, and sexual function.
But fascinating science does not equal safe medicine.
The peptide was abandoned by pharmaceutical companies for good reason. The side effect profile is unacceptable. The long-term safety data does not exist. The risk of serious complications (kidney failure, priapism, cardiovascular events) is real and documented. The melanoma question remains unanswered. And the unregulated market adds an additional layer of danger through contamination and quality control failures.
Regulatory agencies worldwide have issued warnings. Medical professionals have documented harm. And yet, social media continues to promote it as a miracle solution for cosmetic tanning.
If you want tanned skin, use FDA-approved self-tanners. If you want to address sexual dysfunction, consult a physician about evidence-based treatments. And if you're interested in peptide research, work with qualified professionals in controlled settings where safety can be monitored.
No tan is worth risking your kidneys, your heart, or your life.
Interested in FDA-approved peptide therapies? Read about What Are Peptides? The Ultimate Beginner's Guide to Peptide Therapy.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. Melanotan 2 is NOT approved for human use by the FDA, TGA, or any major regulatory body. It is illegal to sell or market for cosmetic or therapeutic purposes in most countries. This article does not endorse or encourage the use of Melanotan 2. Peptides and research compounds are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. The statements regarding these products have not been evaluated by the Food and Drug Administration (FDA). Always consult your local laws and institutional guidelines regarding the use of peptides in research. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay seeking it because of information you read online.