Epithalon: Telomeres and the Science of Life Extension
Epithalon, the only peptide shown to lengthen telomeres and activate telomerase in human cells. Discover the research, anti-aging mechanisms and sourcing guide.
Every cell in your body has a built-in countdown clock.
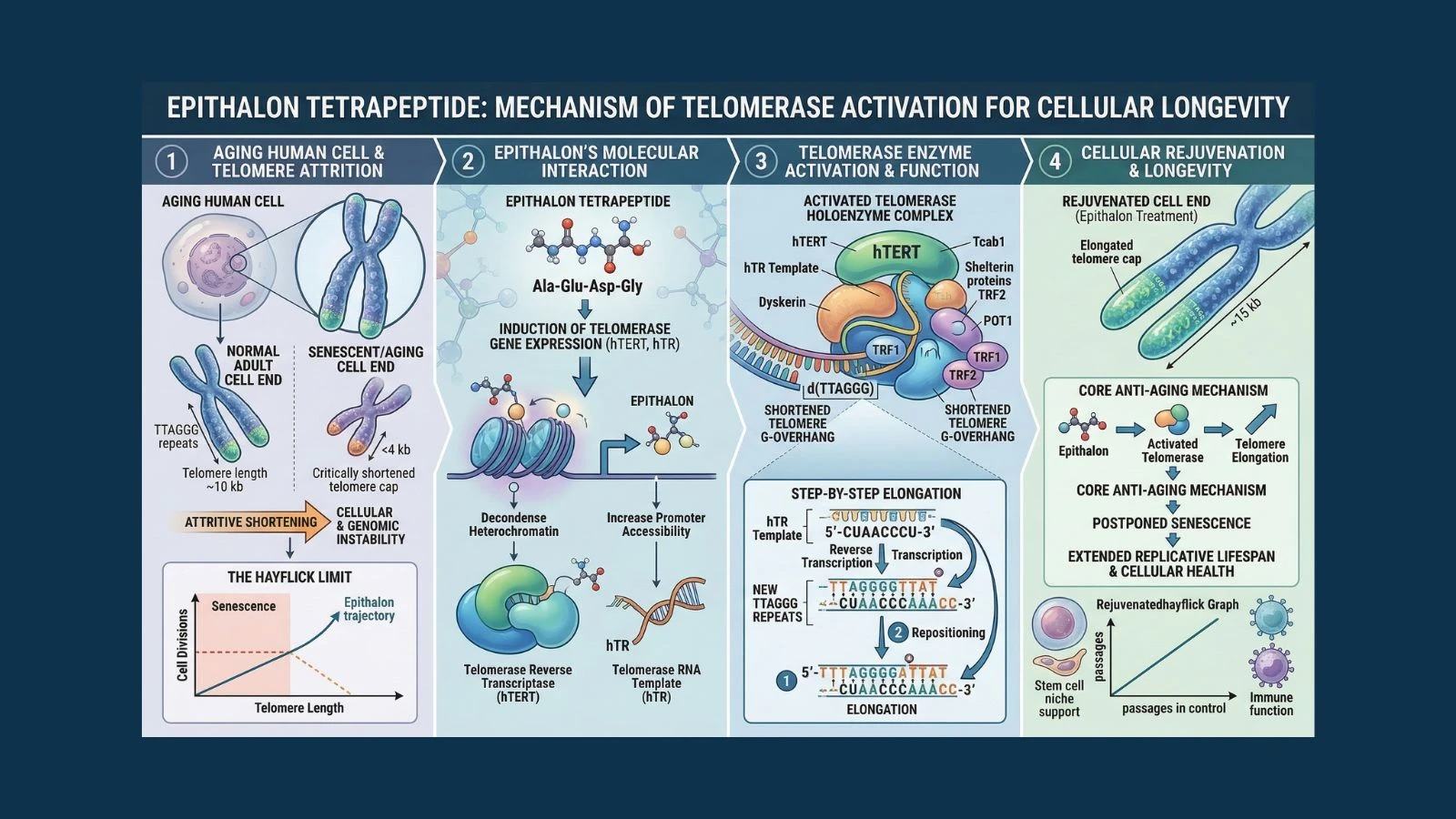
It sits at the very tips of your chromosomes, in structures called telomeres. Every time a cell divides, these protective caps get slightly shorter. When they shrink to a critical length, the cell stops dividing, enters a state called senescence, and begins to contribute to the aging process rather than fighting it. This shortening of telomeres is one of the most fundamental biological mechanisms of aging that science has ever identified.
Epithalon is the only research peptide shown to activate the enzyme that rebuilds these telomere caps and actually lengthen telomeres in human cells. It was developed in Russia over four decades of scientific study and has accumulated one of the most compelling bodies of longevity research of any peptide compound in existence.
For researchers studying aging biology, cellular senescence, longevity science, and the mechanisms of life extension, epithalon stands in a category of its own. This guide covers the full science: what telomeres are, how epithalon activates telomerase, what the decades of Russian research have shown, and why this four-amino-acid peptide has become one of the most talked-about compounds in modern anti-aging science.
New to peptide research? Start with our Complete Beginner's Guide to Peptide Therapy before exploring the deep cellular biology of epithalon.
Understanding Telomeres: The Biological Clock of Aging
Before diving into epithalon itself, it is essential to understand what telomeres are and why they matter so much to the science of aging. Without this foundation, the significance of epithalon's mechanism is easy to miss.
What Are Telomeres?
Telomeres are repetitive DNA sequences that cap the ends of every chromosome in the human body. Think of them like the plastic tips on the ends of shoelaces. The plastic tip does not carry any instruction information, but it protects the actual lace from fraying. Telomeres work the same way: they protect the critical coding DNA within each chromosome from being damaged or degraded during cell division.
In humans, the telomere sequence is a repeating unit of the bases TTAGGG, stacked thousands of times at each chromosome end. A newborn's telomeres might contain 8,000 to 10,000 of these repeating units. By old age, that number has often dropped to just 1,500 to 3,000.
The Hayflick Limit and Cellular Senescence
In 1961, scientist Leonard Hayflick discovered that human cells cannot divide indefinitely. They have a maximum number of divisions, now called the Hayflick limit, typically around 50 to 70 divisions for most human cell types. After that, cells enter senescence, a state where they stop dividing but do not die. Instead, they remain metabolically active and begin secreting inflammatory signals that damage surrounding tissue and accelerate systemic aging.
Telomere shortening is the primary mechanism that enforces the Hayflick limit. Each cell division trims a small section from the telomere ends. When telomeres get too short, they trigger a DNA damage response that puts the cell into permanent senescence. The cell interprets short telomeres as chromosome damage, and it stops dividing as a safety measure.
Research published in PMC (PubMed Central) has confirmed that telomere length is a reliable biomarker of biological age, with shorter telomeres associated with increased risk of age-related disease, reduced immune function, and shorter lifespan across multiple population studies.
What Is Telomerase?
Telomerase is the enzyme responsible for rebuilding telomere length. It adds new TTAGGG repeating units back onto the ends of shortened telomeres, effectively resetting the clock on cell division potential. Most adult somatic cells have very low or inactive telomerase activity, which is why telomeres shorten with age.
Certain cells, including stem cells, immune cells, and reproductive cells, maintain higher telomerase activity to preserve their ability to divide throughout a lifetime. Cancer cells exploit this same mechanism, activating telomerase to achieve unlimited division potential.
The central question in longevity research has always been: can telomerase be safely activated in normal aging cells to slow or reverse telomere shortening without triggering cancer? Epithalon represents one of the most promising answers to that question.
What Is Epithalon? The Pineal Peptide
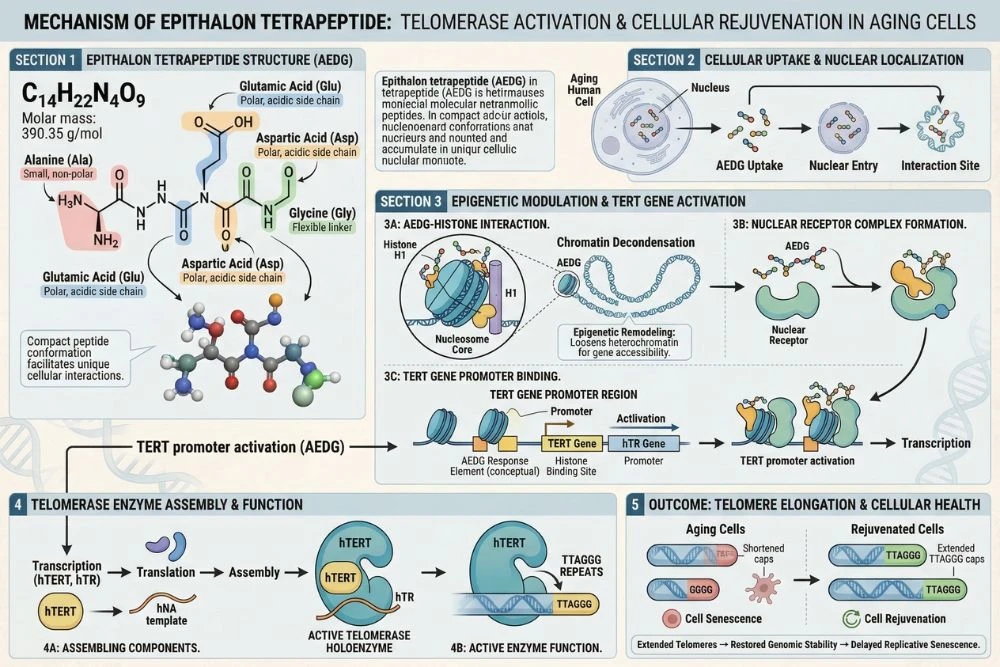
Epithalon is a synthetic tetrapeptide, meaning it is built from just four amino acids: Ala-Glu-Asp-Gly (alanine, glutamic acid, aspartic acid, and glycine). It was developed by Professor Vladimir Khavinson and his team at the St. Petersburg Institute of Bioregulation and Gerontology in Russia, beginning in the 1980s.
Epithalon is the synthetic analog of epithalamin, a natural peptide extract derived from the pineal gland of animals. The pineal gland has long been associated with longevity and biological aging regulation, in large part because it produces melatonin and other regulatory peptides that govern circadian rhythms, immune function, and hormonal balance. Epithalamin was first extracted and studied in the 1970s as part of a broader Soviet research program into bioregulatory peptides.
Professor Khavinson spent decades refining epithalamin into the synthetic tetrapeptide epithalon, which could be precisely manufactured, extensively studied, and eventually published in peer-reviewed scientific literature. The result is a compound with over 100 published studies and one of the longest track records of any longevity-focused research peptide.
Why Just Four Amino Acids?
The simplicity of epithalon is part of what makes it so scientifically interesting. Four amino acids is about as small as a functionally active peptide can get. This tiny size gives epithalon several practical advantages: it is extremely stable compared to larger peptides, it is easy to synthesize with high purity, it can cross biological barriers more easily than large molecules, and it is unlikely to trigger immune responses.
Despite its small size, epithalon's four amino acids fold into a specific three-dimensional shape that allows it to interact with nuclear receptors and gene regulatory regions inside cells, producing effects on gene expression that reach far beyond what its tiny size might suggest.
For a broader look at how small peptide structures produce outsized biological effects, our guide on Simple Peptides: The Scientific Advantage of Short-Chain Research Compounds explores this concept in depth.
How Epithalon Works: Activating Telomerase and Extending Telomeres
Epithalon's mechanism is multi-layered, but the telomerase activation pathway is its most celebrated and scientifically significant effect. Here is how it works step by step.
Step 1: Gene Regulation and Chromatin Remodeling
Epithalon enters the cell nucleus and interacts with chromatin, the complex of DNA and proteins that packages the genome. Specifically, it promotes the decondensation of heterochromatin, which is a tightly packed, transcriptionally inactive form of DNA. By opening up this chromatin structure, epithalon makes previously silenced genes accessible for transcription.
This chromatin remodeling effect is thought to be one of the primary mechanisms through which epithalon produces its wide range of biological effects. Many of the genes that become less active with age are silenced through chromatin compaction. Epithalon partially reverses this age-related gene silencing.
Step 2: Activating the TERT Gene
The most critical target of epithalon's gene regulatory activity is the TERT gene, which encodes for the catalytic subunit of telomerase (telomerase reverse transcriptase). TERT expression is what determines how much telomerase activity a cell has.
In most aging somatic cells, TERT is transcriptionally silenced. The chromatin around the TERT gene is packed tightly, and very little telomerase is produced. Epithalon's chromatin remodeling activity targets the regulatory region of the TERT gene, promoting its expression and leading to increased telomerase production inside the cell.
A landmark study published in PubMed by Khavinson and colleagues demonstrated for the first time that epithalon stimulated telomerase activity in human fetal fibroblast cells, extended their replicative lifespan beyond the Hayflick limit by approximately 10 additional cell divisions, and produced measurable telomere elongation. This was one of the first studies to show that a peptide compound could directly extend cellular lifespan through telomerase activation.
Step 3: Telomere Elongation
With telomerase activated, the enzyme begins its work of adding new TTAGGG repeating units to the ends of shortened telomeres. Cells that were approaching the end of their replicative capacity gain additional division potential. The DNA damage signals that were triggering senescence are reduced as telomeres recover sufficient length.
The result is a cell that has been, in a measurable biological sense, made younger. Its telomeres are longer. Its capacity for further division is restored. The countdown clock has been wound back.
Step 4: Broader Gene Expression Effects
Beyond TERT, epithalon's chromatin remodeling effects alter the expression of dozens of genes involved in aging biology. Research has documented epithalon-induced changes in the expression of genes governing:
Antioxidant defense, with upregulation of superoxide dismutase (SOD) and catalase, two of the body's primary antioxidant enzymes
DNA repair pathways, with increased expression of genes that recognize and fix DNA damage
Apoptosis regulation, reducing inappropriate cell death in healthy tissue while maintaining the ability to eliminate genuinely damaged cells
Inflammatory signaling, with reductions in pro-inflammatory cytokine expression that contribute to chronic age-related inflammation
This broad epigenetic influence on aging-related gene expression is what gives epithalon effects that go well beyond simple telomere management.
The Research Benefits of Epithalon
Four decades of Russian research, combined with more recent international studies, have produced a remarkably detailed picture of epithalon's biological effects. Here is what the evidence shows.
Benefit 1: Telomere Lengthening and Extended Cell Lifespan
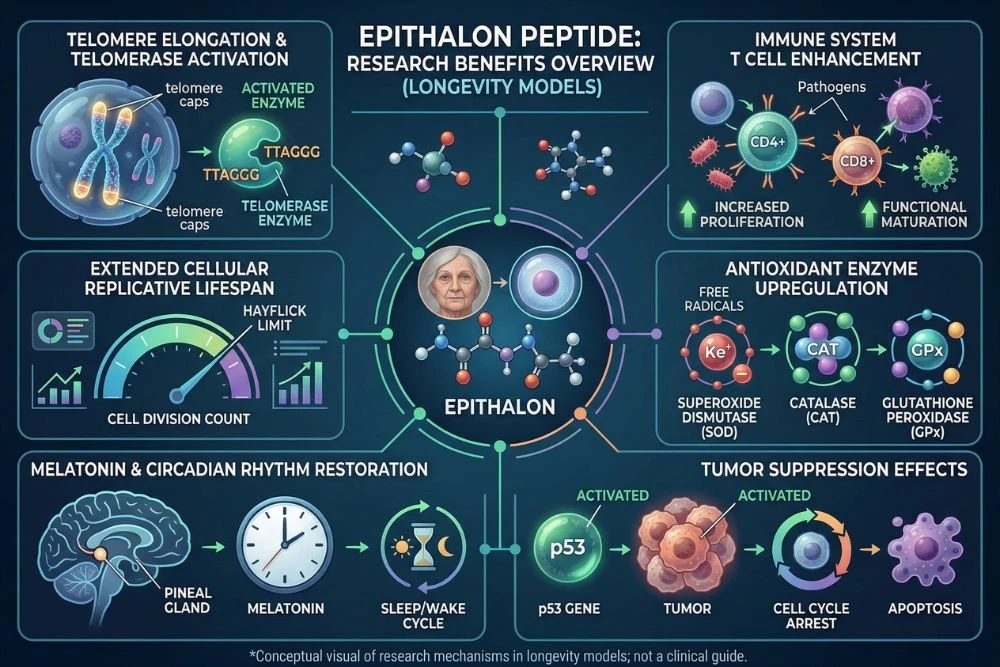
This is epithalon's defining research achievement. The ability to demonstrably lengthen telomeres and extend cellular replicative lifespan in human cell cultures is unmatched by any other peptide compound. Multiple studies have replicated the finding that epithalon treatment produces measurable telomere elongation in human cells, with treated cells outliving their expected Hayflick limit by a statistically significant margin.
Benefit 2: Lifespan Extension in Animal Models
Beyond cell culture studies, epithalon has demonstrated lifespan extension in multiple animal models. Studies in fruit flies, mice, and rats have consistently shown that epithalon-treated animals live longer than untreated controls.
A long-term study published in PMC Gerontology found that epithalon treatment in aging rats reduced mortality, extended mean and maximum lifespan, reduced tumor incidence, and improved several biomarkers of aging including antioxidant enzyme levels and hormonal profiles, providing some of the most comprehensive animal evidence for epithalon's longevity effects.
Benefit 3: Melatonin and Circadian Rhythm Restoration
One of epithalon's most consistently documented effects is the restoration of melatonin production in aging organisms. Melatonin levels decline significantly with age because the pineal gland, which produces melatonin, itself undergoes age-related calcification and reduced function.
Epithalon, as a synthetic analog of a pineal peptide, directly stimulates pineal gland function and restores more youthful melatonin secretion patterns. This has downstream effects on circadian rhythm quality, sleep architecture, immune function, and the antioxidant protection melatonin provides throughout the body.
Restoring melatonin rhythm is considered one of epithalon's most practically significant effects for overall healthspan, since disrupted circadian biology is strongly linked to accelerated aging, immune decline, and metabolic disease.
Benefit 4: Immune System Support
The immune system is one of the earliest systems to show age-related decline. Thymic involution (shrinkage of the thymus gland) begins in early adulthood, reducing the production of naive T cells that are essential for fighting novel pathogens and cancer cells.
Epithalon research has shown improvements in immune function markers, including increased T cell counts and activity, enhanced natural killer (NK) cell function, and improved antibody responses in aging animal models. These immune restoration effects align with the broader pattern of epithalon partially reversing age-related biological decline.
For comparison with another peptide that acts on immune function through a completely different mechanism, our guide on Thymosin Alpha 1: The Gold Standard for Immune Optimization provides essential context on peptide-based immune research.
Benefit 5: Antioxidant and DNA Protection
Oxidative stress is one of the central drivers of cellular aging. Free radicals and reactive oxygen species damage DNA, proteins, and cell membranes, accumulating over time to produce the functional decline we recognize as aging. Epithalon's upregulation of antioxidant enzyme production provides a layer of cellular protection that complements its telomerase activation effects.
Research has also shown that epithalon enhances the expression of DNA repair genes, meaning cells treated with epithalon are better equipped to identify and fix DNA damage before it accumulates into mutations or triggers senescence.
Benefit 6: Cancer Research Implications
In animal cancer models, epithalon has consistently shown tumor-suppressive effects, reducing both the incidence and progression of spontaneous and induced tumors in treated animals. This appears to be related to its immune-enhancing effects, its ability to normalize cell division patterns, and its influence on apoptosis (programmed cell death) in abnormal cells.
Research published in PubMed found that long-term epithalon administration in aging mice significantly reduced the incidence of spontaneous mammary tumors compared to controls, while simultaneously improving survival rates, suggesting a meaningful anti-carcinogenic effect alongside its longevity benefits.
It is important to note that the same telomerase activation that epithalon uses to restore aging cell function is also exploited by cancer cells. This creates a theoretical consideration in cancer research contexts that is discussed in the safety section below.
The Russian Research Legacy: Decades of Epithalon Science
Epithalon's research history is unlike that of any other peptide in this guide series. While most research peptides have a handful of studies from the past decade, epithalon has over 40 years of continuous scientific investigation behind it, the majority conducted at the St. Petersburg Institute of Bioregulation and Gerontology.
Professor Vladimir Khavinson and the Bioregulator Program
Professor Khavinson began his work on bioregulatory peptides in the 1970s, starting with natural peptide extracts from animal organs including the pineal gland, thymus, bone marrow, and retina. The hypothesis was that each organ produces small regulatory peptides that govern its own function and contribute to the body's overall aging rate. By isolating and studying these peptides, and then synthesizing their active fragments, Khavinson's team aimed to understand and eventually slow the aging process.
This program produced dozens of bioregulatory peptide compounds, of which epithalon is the most extensively studied and internationally recognized. Khavinson himself has authored or co-authored hundreds of peer-reviewed publications on epithalon and related compounds, making him one of the most prolific researchers in the field of peptide gerontology.
What the Long-Term Human Studies Show
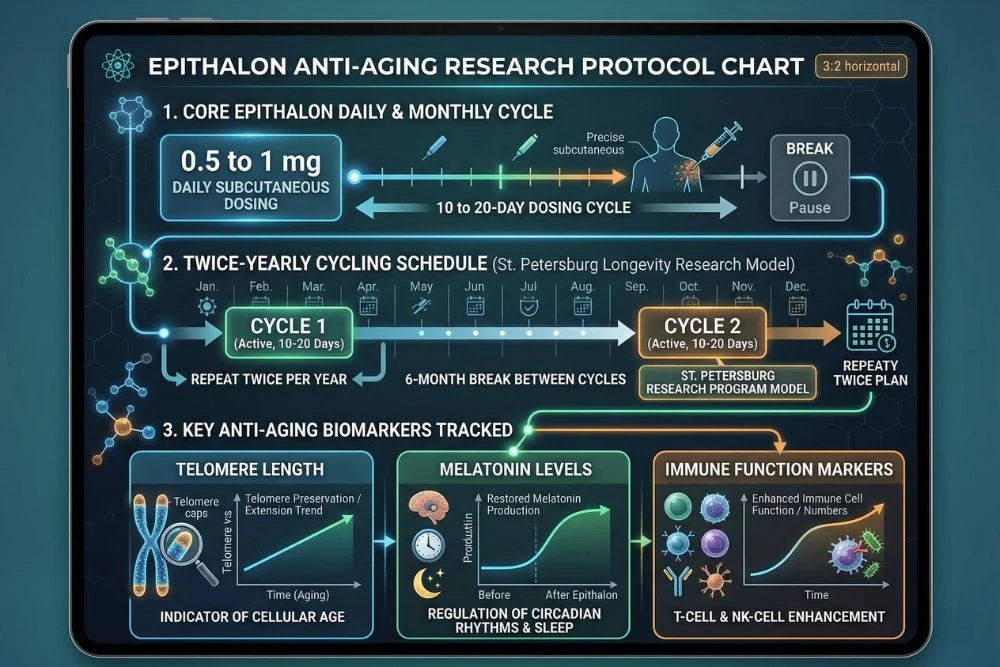
Among the most compelling data from Khavinson's program are the long-term follow-up studies conducted on elderly human participants. These studies tracked groups of elderly patients who received periodic epithalon treatment over years and compared their health outcomes to untreated controls.
A 15-year follow-up study reviewed in PMC Biogerontology found that elderly patients who received periodic epithalon or epithalamin treatment had significantly lower mortality rates, fewer cardiovascular events, and better preservation of cognitive and immune function compared to age-matched controls over the study period. The treated group showed a mortality reduction of approximately 28 percent over 15 years.
While these studies are not randomized controlled trials by modern Western pharmaceutical standards, the scale and duration of the follow-up data is remarkable and represents the kind of long-term outcomes evidence that most research peptides simply do not have.
Research Protocols and Dosing Considerations
Important note: The information below is drawn from published research and clinical use data from the St. Petersburg longevity research program and related studies. Epithalon is a research peptide and is not approved for human use by the FDA or equivalent agencies outside of research settings. This section is for educational and scientific reference only.
Most Commonly Used Research Protocols
Epithalon research protocols vary significantly depending on the study objective, but the most commonly referenced dosing approaches from published literature fall into two categories:
Short course: 5 to 10 mg total across a 10-day cycle, typically administered as 0.5 to 1 mg per day via subcutaneous injection. This type of protocol is used for acute telomerase activation studies and short-term assessment of biological aging markers
Long course: 10 to 20 mg total across a 20-day cycle, with doses of 0.5 to 1 mg per day. This corresponds more closely to the clinical protocols used in the Russian longevity research and is the approach associated with the long-term follow-up outcome data
Cycling and Frequency
In the St. Petersburg research program, epithalon was not administered continuously. Treatment courses were conducted once or twice per year, with several months between cycles. The rationale is that epithalon's epigenetic effects persist well beyond the administration window, and that cycling allows the restored gene expression patterns to stabilize before the next treatment course reinforces them.
The most commonly referenced cycling approach in anti-aging research contexts is two to three treatment courses per year, with each course lasting 10 to 20 days. Some researchers extend to a single longer annual course of 20 days.
Administration Routes
Epithalon is most commonly administered via subcutaneous injection in research protocols. Some research has explored intranasal delivery as a less invasive option. Because epithalon is a small, stable tetrapeptide, it can also survive oral administration to a greater degree than larger peptides, though injectable routes are still considered more reliable for research purposes where consistent bioavailability is important.
Reconstitution and Storage
Epithalon is supplied as lyophilized powder and reconstituted with bacteriostatic water for injectable research use. Reconstituted solutions should be stored at 2 to 8 degrees Celsius. Lyophilized powder is stable at room temperature for short periods but should be refrigerated or frozen for long-term storage.
For step-by-step reconstitution guidance and precise dosing calculations, our Peptide Calculator Guide covers the full process.
Safety Profile and Research Considerations
Epithalon has an exceptionally long and well-documented safety record given its decades of research use. Here is what the data shows.
Observed Tolerability in Research
In published studies and the long-term Russian clinical research program, epithalon has been remarkably well-tolerated. No serious adverse events have been directly attributed to epithalon administration in the published literature across multiple decades of use. Injection site reactions are the most commonly noted observation, consistent with any subcutaneous peptide administration protocol.
Unlike many peptide compounds, epithalon does not interact with the hormonal axis, does not affect GH or sex hormone levels directly, and does not produce the kind of systemic receptor activation that can cause side effects across multiple organ systems. Its effects are primarily at the gene expression level, which produces a gradual and biologically integrated response rather than acute pharmacological effects.
The Telomerase and Cancer Consideration
The most important theoretical safety consideration with epithalon is its telomerase-activating mechanism. Since cancer cells exploit telomerase to achieve unlimited division, the question arises: could activating telomerase in normal cells increase cancer risk?
This is a legitimate research question and one that Khavinson's team took seriously. Notably, the animal studies have consistently shown the opposite of increased cancer risk: epithalon-treated animals demonstrated lower tumor incidence and reduced cancer mortality compared to controls. The proposed explanation is that epithalon's broader effects, including immune enhancement, improved DNA repair, and normalization of apoptosis signaling, create a cellular environment that is better at preventing and eliminating abnormal cell growth even as telomerase activity is moderately elevated.
However, all epithalon research protocols should be designed with appropriate monitoring. Researchers working with subjects who have a history of cancer or pre-cancerous conditions should take this consideration into account in their study design.
Interaction with Other Compounds
Epithalon's mechanism at the gene regulation level means it does not compete with or block the receptors of other research peptides, making it generally combinable with other longevity and performance research compounds without direct pharmacological interaction. The most commonly studied combinations in anti-aging research are epithalon alongside thymosin alpha 1 for immune amplification, and epithalon with GHRPs for a combined cellular rejuvenation and growth hormone axis approach.
Epithalon in the Context of Modern Longevity Science
Epithalon did not emerge from Silicon Valley's longevity labs or from a university biotech spinout. It came from the Soviet Union's biogerontology program, which gave it a very different developmental trajectory from most compounds discussed in Western peptide science circles. Understanding how epithalon fits into the broader modern longevity research landscape helps place its findings in context.
Telomere Research and the 2009 Nobel Prize
In 2009, Elizabeth Blackburn, Carol Greider, and Jack Szostak received the Nobel Prize in Physiology or Medicine for their discovery of telomeres and telomerase. This Nobel recognition brought mainstream scientific attention to the exact biological mechanisms that epithalon research had been targeting for two decades before the prize was awarded. The convergence of the Nobel Prize-recognized science with Khavinson's long-running research program significantly elevated international interest in epithalon.
Epithalon and the Hallmarks of Aging
In 2013, a landmark paper published in Cell identified nine hallmarks of aging, fundamental biological processes whose disruption drives age-related decline. Epithalon's documented mechanisms directly address at least four of these hallmarks:
Telomere attrition: Directly countered through telomerase activation and telomere elongation
Epigenetic alterations: Addressed through chromatin remodeling and restoration of youthful gene expression patterns
Loss of proteostasis: Partially addressed through improved DNA repair and reduced oxidative stress on protein synthesis machinery
Cellular senescence: Reduced through extended replicative lifespan and delayed entry into the senescent state
No other single peptide compound in current research has documented effects on this many hallmarks of aging simultaneously. This breadth of mechanism is what places epithalon at the center of serious longevity research conversations.
Comparing Epithalon to Modern Longevity Compounds
The contemporary longevity research space includes compounds like NAD+ precursors (NMN, NR), rapamycin, metformin, senolytics like dasatinib and quercetin, and gene therapy approaches. Each targets different aging mechanisms. Epithalon's unique advantage is that it is the only compound with direct telomerase activation and telomere elongation evidence in human cells, combined with 40 years of safety and outcomes data that newer compounds simply cannot match.
For a broader perspective on where peptide science is heading in the longevity space, our guide on The Future of Peptide Science: Trends in Regenerative Medicine maps the emerging directions in this rapidly evolving field.
Sourcing Quality Epithalon for Research
Epithalon's simple four-amino-acid structure makes it one of the more straightforward peptides to synthesize, but quality verification remains critical, especially for research where telomerase activation assays and biomarker tracking depend on consistent compound activity.
What a Certificate of Analysis Should Confirm
Quality research-grade epithalon should come with third-party laboratory documentation showing:
HPLC purity at or above 98 percent
Mass spectrometry confirming the correct molecular weight (390.35 Da for Ala-Glu-Asp-Gly)
Amino acid sequence verification of the Ala-Glu-Asp-Gly tetrapeptide
Batch number and certification from an accredited independent laboratory
Our guide on How to Read a Peptide Certificate of Analysis explains every section of a CoA document so you know exactly what to verify before purchasing any research peptide.
Recommended Research Supplier
Peptides Finder connects researchers with vetted suppliers who provide complete third-party testing documentation for epithalon and all major longevity research peptides.
Browse Epithalon Research Compounds at Peptides Finder
We may earn a commission from purchases made through links on this page.
Frequently Asked Questions
What is epithalon and what does it do?
Epithalon is a synthetic tetrapeptide consisting of four amino acids (Ala-Glu-Asp-Gly) that was developed by Professor Vladimir Khavinson at the St. Petersburg Institute of Bioregulation and Gerontology. Its primary and most scientifically significant effect is activating the telomerase enzyme, which rebuilds shortened telomeres on chromosome ends in aging cells. This telomere elongation extends the replicative lifespan of cells beyond their normal Hayflick limit. Epithalon also restores melatonin production, enhances immune function, upregulates antioxidant enzymes, and produces broad epigenetic effects that partially reverse age-related changes in gene expression.
How does epithalon activate telomerase?
Epithalon enters the cell nucleus and promotes the remodeling of chromatin, the packaged structure of DNA and proteins that determines which genes are active or silenced. In aging cells, the TERT gene (which encodes the catalytic subunit of telomerase) is typically silenced by tightly packed heterochromatin. Epithalon's chromatin remodeling effect opens the regulatory region of the TERT gene, promoting its transcription and increasing telomerase enzyme production. With more telomerase active, the enzyme rebuilds the TTAGGG repeat sequences that form telomere caps, effectively restoring telomere length and extending cell division potential.
Is there human evidence for epithalon's longevity effects?
Yes, though the primary human data comes from the Russian research program rather than large Western randomized controlled trials. The most notable human evidence includes a 15-year follow-up study showing that elderly patients who received periodic epithalon or epithalamin treatment had approximately 28 percent lower mortality rates over the study period compared to age-matched untreated controls, with better preservation of immune function and fewer cardiovascular events. Cell culture studies using human cells have also directly demonstrated telomerase activation and telomere elongation in response to epithalon treatment, providing mechanistic confirmation of its biological activity in human tissue.
Does epithalon increase cancer risk?
This is the most common concern raised about epithalon's telomerase-activating mechanism. Cancer cells exploit telomerase to divide indefinitely, so there is a theoretical question of whether activating telomerase in normal cells could increase cancer risk. However, the animal research data, which spans decades and multiple species, consistently shows the opposite: epithalon-treated animals have lower tumor incidence and reduced cancer mortality compared to untreated controls. The proposed explanation is that epithalon's complementary effects, including immune enhancement and improved DNA repair, create a cellular environment that suppresses abnormal cell growth. That said, all epithalon research protocols involving subjects with cancer history should include appropriate monitoring and be designed with this consideration in mind.
What is the standard research dosing protocol for epithalon?
The most commonly referenced research protocols use 0.5 to 1 mg of epithalon per day administered via subcutaneous injection, for a course of 10 to 20 days. This is typically cycled two to three times per year with several months between courses, following the approach used in the St. Petersburg longevity research program. Total course doses range from 5 to 20 mg depending on the protocol. The cycling approach is important because epithalon's epigenetic effects on gene expression persist after the administration course ends, and spacing courses allows those changes to stabilize before reinforcement.
How does epithalon compare to other anti-aging compounds?
Epithalon occupies a unique position in the anti-aging research landscape because it is the only compound with direct telomerase activation and telomere elongation evidence in human cells, backed by over 40 years of research data. Most modern longevity compounds like NAD+ precursors, rapamycin, and senolytics target different aging mechanisms such as energy metabolism, mTOR signaling, or senescent cell clearance. Epithalon's documented effects span multiple hallmarks of aging simultaneously, including telomere attrition, epigenetic alterations, and cellular senescence, giving it a breadth of mechanism that few single compounds can match. Its long safety record is also a meaningful advantage over newer compounds with limited long-term data.
Can epithalon be combined with other research peptides?
Yes. Epithalon operates primarily through gene regulation and epigenetic mechanisms rather than through receptor competition, which means it generally does not interfere with the mechanisms of other research peptides. The most commonly studied combinations in longevity research are epithalon alongside thymosin alpha 1 for amplified immune restoration, and epithalon with GHRP compounds for a combined cellular rejuvenation and growth hormone axis approach. Some researchers also combine epithalon with BPC-157 or other tissue repair peptides as part of comprehensive anti-aging protocols, though specific combination data is more limited than single-compound research.
Conclusion: Epithalon and the Promise of Telomere-Based Longevity
Few compounds in all of peptide science carry the scientific weight and historical depth of epithalon. Built from just four amino acids, studied for over four decades, and validated by the same biological mechanisms that earned a Nobel Prize in 2009, epithalon represents the closest thing science currently has to a direct molecular intervention on the aging clock.
The telomere shortening that drives cellular senescence, tissue decline, and age-related disease is not inevitable in the way scientists once believed. Epithalon's ability to activate telomerase, rebuild shortened telomeres, restore youthful gene expression patterns, and extend cellular lifespan makes it a fundamentally different kind of research compound than anything else in the peptide world.
The long-term human follow-up data from the St. Petersburg research program adds a dimension of real-world outcomes evidence that most research peptides will never have. Thirty percent lower mortality over 15 years is not a cell culture finding. It is a signal that something meaningful is happening at the biological level.
For researchers serious about longevity, aging biology, cellular rejuvenation, and the science of life extension, epithalon is not just one option among many. It is the benchmark against which every other anti-aging intervention should be measured.
Explore the full library of anti-aging, immune, and longevity peptide research guides at Peptides Finder, and stay current with the latest developments in telomere science and life extension research.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. Epithalon is sold as a research chemical. Research peptides are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any research compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.