DSIP: Understanding the Deep Sleep Inducing Peptide
DSIP peptide promotes deep slow-wave sleep by modulating stress hormones and brain oscillations. Discover the research, dosing protocols, and benefits for 2026.
Sleep is not passive. It is one of the most metabolically active and biologically productive states the human body enters.
During deep sleep, the brain clears waste products through the glymphatic system, the pituitary releases the majority of its daily growth hormone pulse, muscles and connective tissue are rebuilt, and memories are consolidated from short-term storage into long-term archives. The quality of your deep sleep determines the quality of your recovery, your hormonal balance, your cognitive performance the next day, and over a lifetime, your resistance to neurodegenerative disease.
DSIP, the delta sleep-inducing peptide, is the only known endogenous peptide that directly promotes the deepest stage of sleep: slow-wave sleep, also called delta sleep. It was discovered in 1974 when researchers isolated it from the brain fluid of sleeping rabbits and found it could transfer the deep sleep state to awake animals when injected. That discovery launched five decades of research into one of the most fascinating and complex compounds in sleep science.
This guide covers the full story of DSIP: what it is, how it works, what the research shows about its effects on sleep architecture and stress biology, and why it continues to be one of the most studied neuropeptides in sleep research despite questions that remain open about its exact mechanism.
New to research peptides? Our Complete Beginner's Guide to Peptide Therapy gives you the scientific foundation you need before exploring the neurobiology of DSIP.
What Is DSIP? The Discovery That Changed Sleep Research
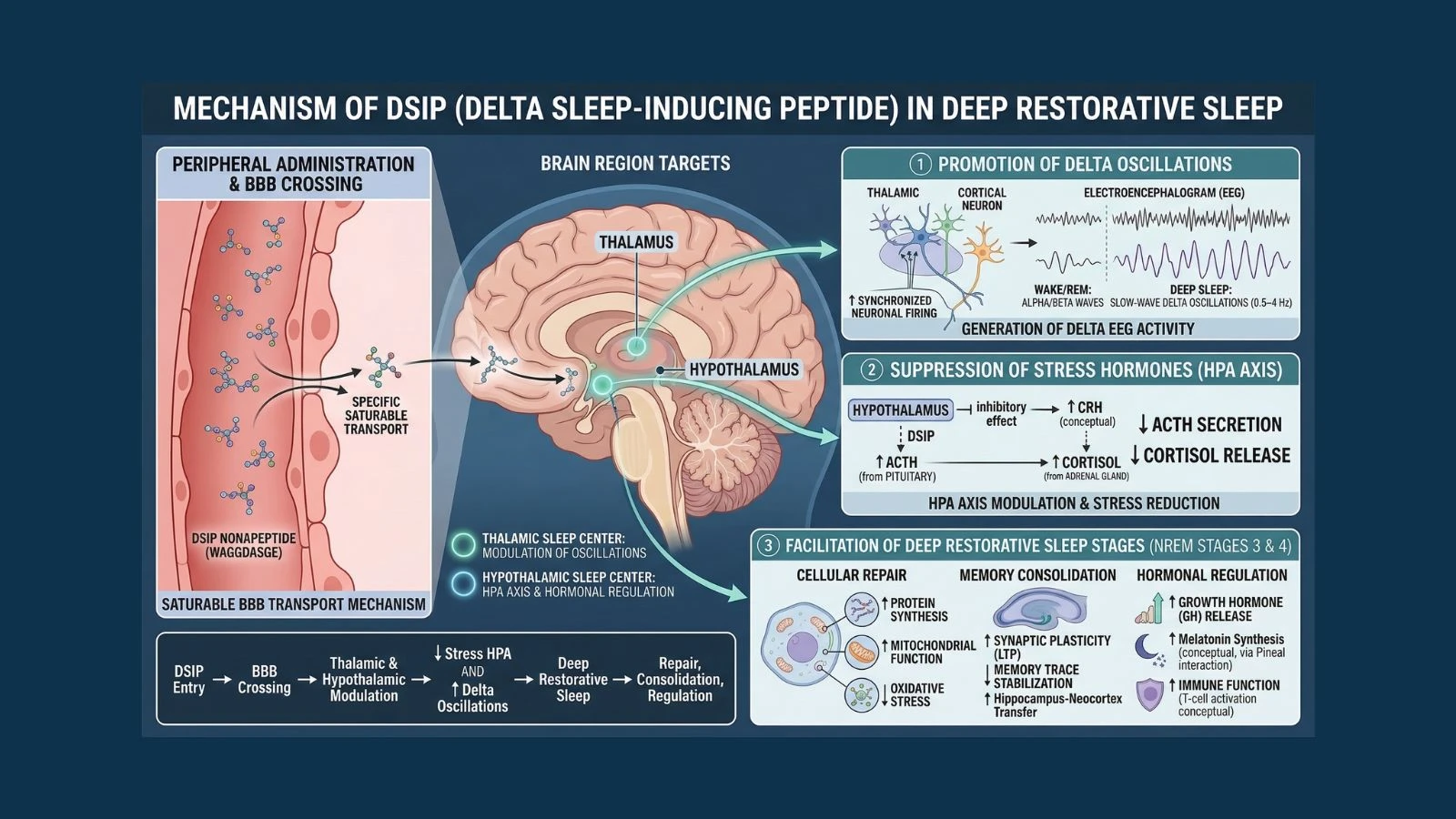
DSIP stands for Delta Sleep-Inducing Peptide. It is a naturally occurring neuropeptide consisting of nine amino acids: Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu. It was first isolated in 1974 by Marcel Monnier and his team at the University of Basel in Switzerland.
The discovery was made through a now-classic experiment. Researchers surgically connected the circulatory systems of two rabbits so that blood could flow between them. They then electrically stimulated the thalamus of one rabbit to induce deep sleep. The sleeping rabbit's blood was collected and injected into an awake rabbit. The awake rabbit fell into the same deep, slow-wave sleep pattern.
The conclusion was unmistakable: something in the blood of the sleeping rabbit was carrying a chemical signal for deep sleep. After extensive biochemical analysis, Monnier's team isolated the nine-amino-acid peptide responsible and named it for the type of sleep it induced: delta sleep-inducing peptide.
This was one of the first demonstrations that sleep could be transmitted chemically, and it opened an entirely new line of investigation into the molecular basis of sleep regulation.
DSIP in the Human Body
DSIP is produced naturally in the hypothalamus and has been found in the pituitary gland, blood, cerebrospinal fluid, and peripheral tissues including the gut, pancreas, and adrenal glands. Its widespread distribution across tissues beyond the brain suggested from early on that DSIP was not simply a sleep molecule but a multifunctional regulatory peptide with roles throughout the body.
DSIP crosses the blood-brain barrier efficiently, which is unusual for peptides of even its small size. This ability to move freely between the bloodstream and brain tissue is one of the reasons it can produce central nervous system effects even when administered peripherally, and it is a key practical advantage for research administration.
Research published in PubMed confirmed DSIP's presence in human blood and cerebrospinal fluid with concentrations that fluctuate in a pattern correlated with sleep-wake cycles, providing early evidence that DSIP functions as a genuine endogenous sleep-regulatory signal rather than an experimental artifact.
The Naming Question: Does It Really Induce Delta Sleep?
It is worth addressing a nuance that often creates confusion in DSIP research: the compound's name describes its most famous effect, but the full picture of how it produces that effect is still being worked out. DSIP does not appear to bind to a single identified “sleep receptor.” Instead, it modulates multiple neurological and endocrine systems that collectively create the conditions for deep sleep.
This multi-system, indirect mechanism is actually consistent with how the brain manages sleep more broadly. Sleep is not switched on by one molecule. It is an emergent state that arises when multiple systems align: cortisol drops, GABA activity rises, thalamic oscillations shift to slower frequencies, body temperature falls, and adenosine accumulates. DSIP influences several of these simultaneously.
How DSIP Works: The Multi-System Sleep Mechanism
DSIP produces its sleep-promoting and stress-modulating effects through several interconnected biological pathways. Here is what the research has established.
Suppression of the HPA Stress Axis
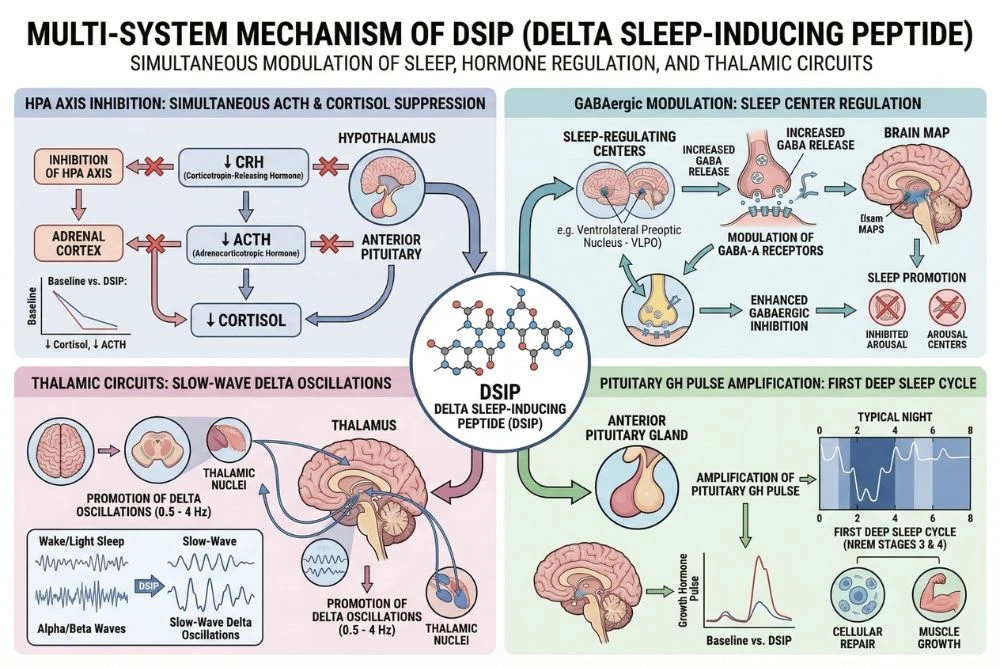
The hypothalamic-pituitary-adrenal (HPA) axis is the body's primary stress response system. When the brain perceives threat or stress, the hypothalamus releases CRH (corticotropin-releasing hormone), which signals the pituitary to release ACTH (adrenocorticotropic hormone), which then signals the adrenal glands to produce cortisol.
Elevated cortisol is one of the most powerful inhibitors of deep sleep. It keeps the brain in a state of elevated alertness, suppresses the slow-wave oscillations characteristic of delta sleep, and fragments sleep architecture. This is why people under chronic stress consistently report poor sleep quality even when they manage to fall asleep.
DSIP directly inhibits the release of ACTH from the pituitary, reducing the downstream cortisol signal. By dampening the HPA axis, DSIP removes one of the main biological barriers to entering and maintaining deep sleep.
Research published in PubMed confirmed DSIP's ability to suppress ACTH-stimulated cortisol secretion, demonstrating that its sleep-promoting effects are partly mediated through direct inhibition of the stress hormone cascade that normally opposes restorative sleep.
Modulation of Thalamic and Hypothalamic Sleep Centers
The thalamus is the brain's relay station for sensory information and plays a central role in generating the slow oscillatory patterns associated with deep sleep. During slow-wave sleep, thalamic neurons fire in synchronized bursts at 0.5 to 4 Hz (delta frequencies), creating the characteristic delta waves visible on EEG recordings. These oscillations are what define the deepest and most restorative stage of sleep.
DSIP modulates neuronal activity in both the thalamus and hypothalamus in ways that promote the transition to and maintenance of these slow oscillatory states. The hypothalamus, which contains the master biological clock (the suprachiasmatic nucleus) and the sleep-promoting ventrolateral preoptic area (VLPO), is particularly sensitive to DSIP's regulatory influence.
GABAergic Modulation
Like several other sleep-relevant compounds, DSIP influences GABAergic signaling, the brain's primary inhibitory neurotransmitter system. GABA activity is essential for the transition from wakefulness to sleep, and the progressive increase in GABAergic tone that occurs as sleep deepens is a fundamental mechanism of sleep architecture.
DSIP's enhancement of GABAergic activity in sleep-relevant brain regions contributes to the reduction in neural excitability needed for slow-wave sleep to establish and maintain itself. This is complementary to its HPA axis suppression: together, they address both the neurological excitability and the hormonal barriers to deep sleep.
Growth Hormone Release Promotion
One of DSIP's most practically significant effects for researchers studying recovery and longevity is its ability to promote growth hormone (GH) release. The largest pulse of GH in a 24-hour period occurs during the first deep sleep cycle of the night, specifically during slow-wave sleep. This GH pulse drives tissue repair, muscle protein synthesis, fat metabolism, and immune function.
By deepening and extending slow-wave sleep, DSIP directly amplifies this nighttime GH pulse. Research has shown that DSIP administration increases both the depth and duration of slow-wave sleep, which corresponds to a larger and more sustained GH release during the critical first sleep cycle.
For researchers studying the growth hormone axis and peptides that work through GH-stimulating mechanisms, our guide on Hexarelin: The Strongest GHRP for Mass and Strength provides comprehensive context on the GH release mechanisms that DSIP's sleep improvement supports.
Antioxidant and Neuroprotective Properties
Research has revealed that DSIP has antioxidant properties that extend beyond its sleep effects. DSIP has been shown to reduce oxidative stress markers in neural tissue, potentially contributing to the neuroprotective effects of healthy deep sleep. The brain's primary waste clearance system, the glymphatic system, operates most actively during slow-wave sleep, and by deepening this sleep stage, DSIP may amplify the brain's nightly maintenance and detoxification processes.
Sleep Architecture: Why Deep Sleep Is the Most Important Stage
To understand why DSIP research matters, you need to understand what sleep actually is and why the deep stage DSIP promotes is the one that most people are chronically missing.
The Sleep Cycle and Its Stages
Human sleep cycles through four stages approximately every 90 minutes throughout the night. The first three stages are non-REM (NREM) sleep, and the fourth is REM sleep. Within NREM sleep, stages 3 and 4 represent slow-wave sleep, the deep delta sleep that DSIP promotes.
- Stage 1 (NREM 1): Light sleep, the transition from wakefulness. Easy to wake from. Takes up about 5 percent of total sleep time
- Stage 2 (NREM 2): Deeper light sleep. Heart rate and body temperature drop. Sleep spindles appear on EEG. Takes up about 45 to 55 percent of total sleep time
- Stage 3 (NREM 3, Slow-Wave Sleep): Deep delta sleep. The brain produces large, slow delta waves. This is the most physically restorative stage. Growth hormone is released here. Takes up 15 to 25 percent of total sleep in healthy young adults but declines significantly with age and stress
- REM Sleep: Rapid eye movement sleep. The brain is highly active. Dreams occur. Critical for emotional processing and memory consolidation. Takes up about 20 to 25 percent of total sleep time
What Happens During Slow-Wave Sleep
Stage 3 slow-wave sleep is where the most critical biological restoration happens:
- The pituitary releases 70 to 80 percent of its daily growth hormone output, driving muscle repair, fat metabolism, and immune function
- The glymphatic system of the brain operates at peak efficiency, clearing metabolic waste products including amyloid beta, a protein associated with Alzheimer's disease when it accumulates
- Cortisol reaches its daily nadir, providing the lowest inflammatory and catabolic environment of the 24-hour cycle
- The immune system conducts surveillance and repair, with natural killer cell activity and T cell function at their nightly peak
- Memory consolidation transfers declarative memories from the hippocampus to long-term cortical storage
Why Modern Lifestyles Destroy Deep Sleep
Slow-wave sleep is the most fragile stage of sleep. It is the first to be reduced by stress, alcohol, artificial light exposure, late-night eating, irregular sleep schedules, and the natural aging process. After age 30, slow-wave sleep begins a steady decline. By age 60, many people get almost no slow-wave sleep at all.
This decline in deep sleep is directly correlated with the health decline that accompanies aging: reduced GH output, increased cortisol, impaired immune function, cognitive decline, and slower physical recovery. Addressing slow-wave sleep is one of the most impactful interventions available for overall healthspan, which is exactly why DSIP research has attracted sustained attention from longevity scientists.
The connection between sleep quality, GH output, and cellular aging is also explored in our guide on Epithalon: Telomeres and the Science of Life Extension, which covers how nighttime hormonal patterns intersect with cellular aging mechanisms.
The Research Benefits of DSIP
Five decades of research have documented DSIP's biological effects across multiple domains. Here is what the evidence shows.
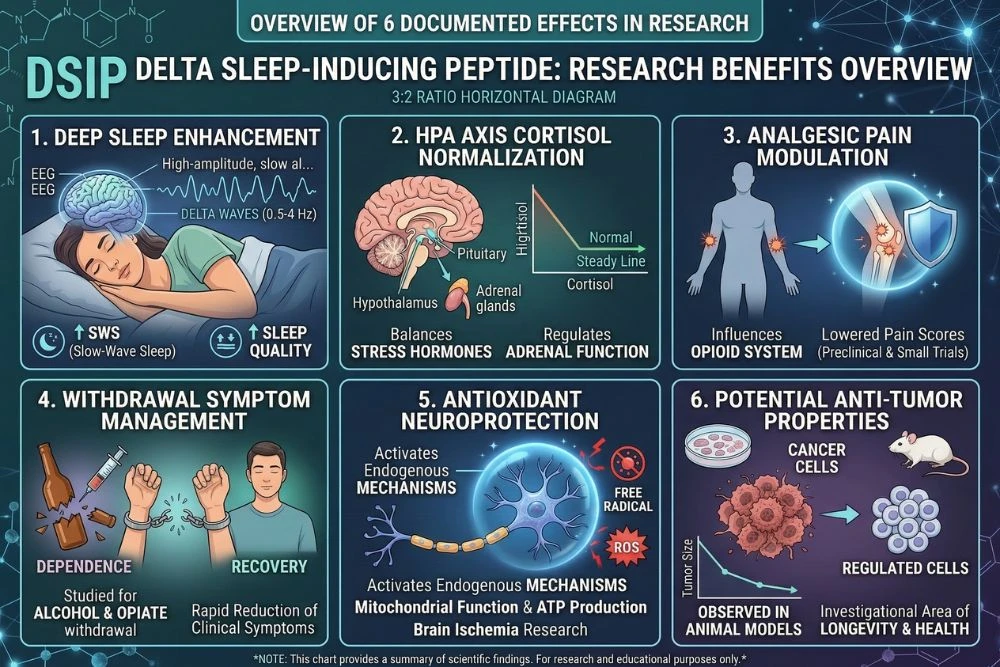
Benefit 1: Increased Slow-Wave Sleep Duration and Quality
This is DSIP's primary and best-documented research effect. Multiple studies across animal models and early human trials have shown that DSIP administration increases the amount of time spent in slow-wave sleep, reduces sleep onset latency (how long it takes to fall asleep), and reduces sleep fragmentation during the night.
A foundational study published in PubMed confirmed that DSIP injections in rabbits produced clear increases in delta sleep measured by EEG, with delta wave amplitude and duration both significantly elevated compared to controls, replicating the original 1974 discovery with more rigorous electrophysiological measurement.
In human studies, DSIP has demonstrated sleep-promoting effects in both healthy volunteers and in patients with insomnia. Participants report falling asleep faster, experiencing more continuous sleep, and feeling more rested the following morning.
Benefit 2: Stress Hormone Normalization
DSIP's inhibition of the HPA axis produces meaningful reductions in cortisol and ACTH levels during the critical nighttime window when these hormones need to be at their lowest for restorative sleep to occur. Research subjects with elevated nighttime cortisol, a common finding in insomnia and chronic stress, show normalization of cortisol profiles following DSIP treatment.
This stress hormone normalization effect extends beyond just sleep improvement. Lower overnight cortisol means less overnight catabolism (breakdown of muscle tissue), better immune function during sleep, and lower inflammatory activity throughout the night. The downstream health implications of normalized nighttime cortisol are significant across multiple biological systems.
Benefit 3: Pain Modulation
An unexpected but repeatedly documented effect of DSIP is pain modulation. DSIP has demonstrated analgesic properties in animal models, reducing pain sensitivity through mechanisms that appear to involve both the opioid system and other neuropeptide pathways.
Research published in PubMed showed that DSIP produced significant analgesia in rodent pain models, with effects comparable to low-dose opioids but through a mechanism that did not involve direct opioid receptor binding, suggesting DSIP activates a distinct pain-modulatory pathway relevant to understanding chronic pain and sleep disruption research.
The relationship between pain and sleep is bidirectional: poor sleep amplifies pain sensitivity, and chronic pain disrupts sleep. DSIP's ability to address both sides of this cycle simultaneously makes it a particularly interesting compound for research into chronic pain conditions where sleep disruption is a major comorbidity.
Benefit 4: Withdrawal and Stress Management
DSIP has been studied in the context of opioid, benzodiazepine, and alcohol withdrawal, where its HPA axis suppression and sleep-promoting effects are particularly relevant. Withdrawal from these substances is characterized by hyperactivation of the stress response, severe sleep disruption, and anxiety, all of which DSIP directly addresses.
Animal models of opioid withdrawal have shown that DSIP reduces withdrawal-associated behavioral signs and normalizes sleep architecture during the acute withdrawal period. This positions DSIP as a research tool for addiction medicine studies at the intersection of sleep science and dependency biology.
Benefit 5: Antioxidant and Neuroprotection
Beyond its sleep and stress effects, DSIP has demonstrated antioxidant activity in neural tissue, reducing markers of oxidative stress in brain cells. Given that oxidative stress is one of the primary drivers of neurodegeneration, and that deep sleep is the primary window for the brain's antioxidant and waste-clearance processes, DSIP's combination of sleep promotion and direct antioxidant activity creates a potentially synergistic neuroprotective effect.
For researchers studying the intersection of sleep, oxidative stress, and cellular aging, our guide on Epithalon: Telomeres and the Science of Life Extension covers how restoring youthful sleep patterns connects directly to cellular longevity mechanisms.
Benefit 6: Potential Anti-Tumor Properties
Like several other neuropeptides with broad biological activity, DSIP has been studied in cancer biology contexts. Research in animal tumor models has shown DSIP can inhibit tumor growth and reduce metastasis through mechanisms related to its stress hormone suppression and immune modulation. Chronic elevated cortisol is known to impair immune surveillance of cancer cells, so DSIP's cortisol-lowering effects may contribute to better immune oversight of abnormal cell growth.
This is an early-stage research area with animal model data rather than human clinical evidence, but it adds another dimension to DSIP's biological profile that researchers across multiple fields have found relevant.
DSIP and the Science of Sleep Research: What Remains Unanswered
Intellectual honesty is important in any scientific discussion of DSIP. Despite five decades of research, some fundamental questions about how DSIP produces its effects remain open. Understanding these open questions is important for researchers designing protocols with DSIP.
The Receptor Question
Unlike many research peptides that act through a well-characterized single receptor, DSIP's specific receptor or receptors have not been definitively identified. The compound clearly produces biological effects, but the exact molecular docking mechanism is still a subject of active research. Some studies suggest DSIP may act through opioid receptors, others through distinct peptide receptors not yet fully characterized, and others suggest its effects are mediated indirectly through the systems it regulates (HPA axis, GABA, etc.) rather than through a primary receptor.
This is not a reason to dismiss DSIP's documented effects. Many biologically active compounds were used effectively for decades before their full receptor pharmacology was understood. Aspirin's mechanism was not fully characterized until nearly a century after its clinical use began. What matters for research is that the biological effects are reproducible and the compound is safe, both of which DSIP has demonstrated consistently.
Variability in Human Response
Human studies with DSIP have shown more variability in response than animal studies. Some human subjects show strong sleep-promoting effects, while others show modest or minimal responses. This variability may reflect individual differences in baseline sleep architecture, stress hormone levels, DSIP receptor sensitivity, or other factors not yet fully understood.
For researchers designing human DSIP studies, this variability means that response measurement needs to be built into the protocol from the outset, and that population stratification by baseline sleep quality may be important for detecting meaningful effects.
Half-Life and Stability
DSIP has a relatively short half-life in peripheral circulation due to enzymatic degradation, typically estimated at 30 to 60 minutes in blood. This short half-life creates questions about how a single injection produces sleep effects that last for a full night. Some researchers propose that DSIP acts as a trigger rather than a sustained signal, activating systems that then maintain their own momentum independently. Others suggest that DSIP's effects in the brain outlast its presence in the bloodstream because it triggers durable changes in neurotransmitter levels rather than requiring continuous receptor occupancy.
This mechanistic uncertainty is one of the genuinely interesting open questions in DSIP research and represents an active area of investigation.
Research Protocols and Dosing Considerations
Important note: The following information is drawn from published research studies conducted in controlled laboratory and clinical settings. DSIP is a research peptide and is not approved for human use by the FDA. This section is for educational and scientific reference only.
Dosing Ranges in Published Research
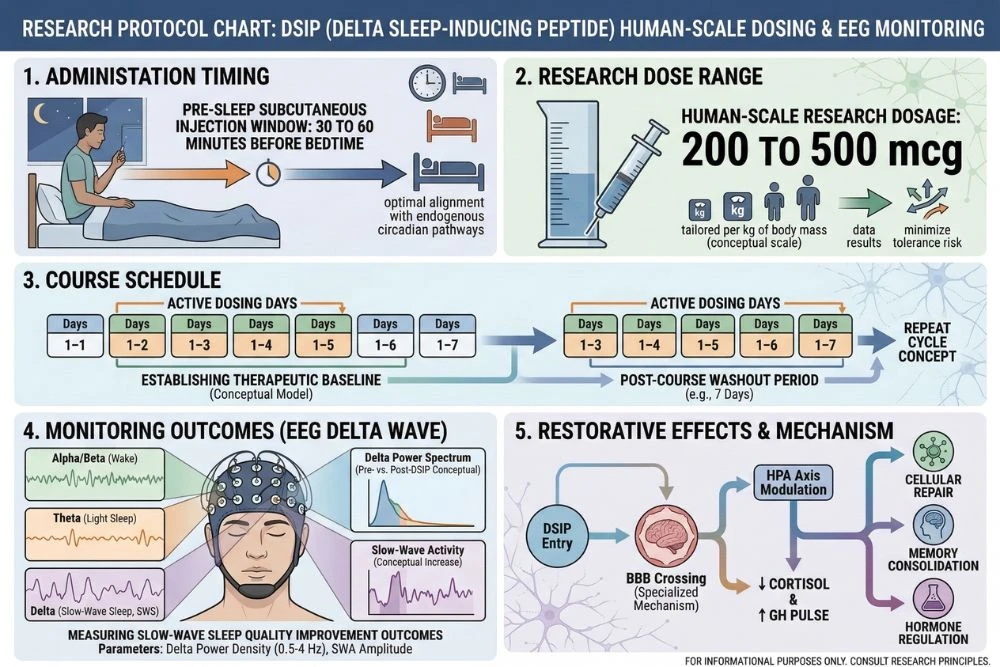
DSIP has been administered at a range of doses in published studies, with the following ranges most commonly referenced:
- Low range: 25 to 50 mcg per kilogram of body weight, used in dose-finding studies and sensitivity assessments in animal models
- Human clinical research range: 10 to 40 nanomoles per kilogram, which translates to approximately 200 to 600 mcg total dose for a typical adult research subject
- Practical research dosing: Most published human-context research has used single doses in the 200 to 500 mcg range administered subcutaneously or intravenously
Timing of Administration
Because DSIP's primary research application is sleep promotion, administration timing is critical. Published protocols consistently administer DSIP in the evening, typically 30 to 60 minutes before the intended sleep time. This aligns with DSIP's onset of action and its role in initiating rather than maintaining sleep.
Some research protocols have also studied DSIP administered earlier in the afternoon to assess whether earlier priming of the sleep systems produces different outcomes than immediate pre-sleep administration, but the pre-sleep window remains the most studied and most consistent timing approach.
Subcutaneous vs. Intravenous Administration
Both subcutaneous and intravenous administration routes have been used in DSIP research. Intravenous administration was used in many of the early clinical studies and produces more immediate and predictable plasma levels. Subcutaneous injection is more practical for outpatient research settings and has also been shown to produce meaningful sleep-promoting effects, though with slightly slower onset.
Given DSIP's ability to cross the blood-brain barrier efficiently, subcutaneous administration is generally adequate for most research purposes and is the preferred route in contemporary protocols.
Cycling and Frequency
Unlike compounds where tolerance or receptor desensitization is a concern, published DSIP research does not document significant tolerance development with repeated use. Some protocols have used DSIP on consecutive nights for 5 to 7 day courses, others on a several-times-per-week basis, and others as needed for specific sleep disruption scenarios. The available data does not suggest that continuous use produces diminishing returns, though most published protocols use DSIP in defined courses rather than indefinitely.
Reconstitution and Storage
DSIP is supplied as lyophilized powder for research use and reconstituted with bacteriostatic water. Reconstituted solutions should be refrigerated at 2 to 8 degrees Celsius. The peptide is relatively stable once reconstituted compared to some larger peptides, but protection from light and temperature fluctuation is still important for maintaining activity.
For complete reconstitution instructions and accurate dosing calculations, our Peptide Calculator Guide walks through the full process for any research peptide.
Safety Profile and Research Considerations
DSIP's safety profile across five decades of research is reassuring. Here is what the published data shows.
Reported Side Effects
In published clinical and research studies, DSIP has been well-tolerated with a minimal side effect profile. The most commonly reported observations include:
- Brief sensation of warmth or mild flushing immediately after injection, typically resolving within 10 to 15 minutes
- Mild injection site reactions consistent with any subcutaneous peptide administration
- In a small subset of subjects, a brief period of unusual dreams during early sleep stages, thought to reflect the altered sleep architecture rather than an adverse effect
No serious adverse events have been reported in the published DSIP literature at standard research doses. Importantly, DSIP does not produce the morning grogginess (sleep inertia) associated with many sleep medications, including benzodiazepines and Z-drugs like zolpidem. This is consistent with its mechanism of deepening natural sleep rather than sedating the brain pharmacologically.
No Dependence or Tolerance
DSIP does not act on benzodiazepine or opioid receptors in the way that sleep medications and pain medications do, and published research has not documented physical dependence or tolerance with DSIP use. This is a meaningful safety advantage over conventional pharmacological sleep interventions, where dependence and rebound insomnia are clinically significant concerns with extended use.
Interaction with Sleep Medications
Researchers combining DSIP with other GABAergic or sedating compounds should be aware of potential additive sedation effects. DSIP's GABAergic modulation means it could produce stronger-than-expected sedation when combined with benzodiazepines, Z-drugs, alcohol, or other CNS depressants. Research protocols that include DSIP should account for this and avoid unintended compound interactions.
Considerations for Specific Populations
DSIP's HPA axis suppression effects are generally beneficial for people with chronically elevated cortisol, but researchers working with subjects who have compromised adrenal function should monitor cortisol levels appropriately. In subjects where the adrenal axis is already suppressed (for example, those on long-term corticosteroid therapy), DSIP's additional ACTH inhibition should be a consideration in study design.
DSIP in Context: Comparing Sleep Peptide Research Approaches
DSIP is not the only research compound studied for sleep improvement. Understanding where it sits relative to other approaches helps researchers select the right tool for their specific study question.
DSIP vs. Melatonin
Melatonin is the most widely studied sleep-regulating compound in the world and the most commonly used sleep supplement. It primarily regulates circadian timing, signaling to the brain that it is night and initiating the cascade of events that lead to sleep. Melatonin does not directly promote slow-wave sleep or suppress cortisol to the degree DSIP does. For research into circadian rhythm regulation, melatonin is the appropriate tool. For research specifically targeting slow-wave sleep depth and stress hormone normalization, DSIP addresses mechanisms melatonin does not.
DSIP vs. Selank
Selank, covered in our previous guide, reduces anxiety and promotes GABAergic calm through a daytime nootropic mechanism. It can improve sleep quality indirectly by reducing the anxiety and stress activation that prevent deep sleep. DSIP is more directly targeted at the sleep architecture itself, working through specific sleep-regulatory brain centers rather than through general anxiety reduction. Both compounds address the stress-sleep axis but from different directions and with different primary applications.
For the full picture on selank's anxiety and cognitive mechanisms, our guide on Selank: The Nootropic Peptide for Anxiety and Focus provides the complementary context for understanding how these two neuropeptides can work together in a comprehensive neurological research stack.
DSIP vs. Epithalon
Epithalon promotes deep sleep as part of its broader anti-aging mechanism through pineal gland activation and melatonin restoration. DSIP acts more directly on the acute sleep-promoting system, specifically targeting the slow-wave sleep machinery. The two compounds address sleep at different levels and timescales: DSIP produces more immediate sleep architecture improvements, while epithalon's sleep benefits emerge as part of a slower restoration of the pineal-melatonin system. Combining both in longevity research protocols addresses sleep promotion through complementary mechanisms simultaneously.
DSIP vs. Growth Hormone Secretagogues
GHRPs like hexarelin and ipamorelin stimulate GH release directly from the pituitary. DSIP promotes GH release indirectly by deepening the slow-wave sleep during which the pituitary's largest natural GH pulse occurs. Both approaches increase nighttime GH output, but through completely different routes. DSIP's approach preserves the natural sleep-GH coupling that produces the most biologically complete hormonal cascade, while direct GHRPs can produce GH spikes independent of sleep stage. For research into natural sleep-associated GH optimization, DSIP is the more physiologically authentic tool.
Sourcing Quality DSIP for Research
DSIP's nine-amino-acid sequence is well-characterized, and quality verification through standard analytical methods is straightforward. As with all research peptides, third-party testing documentation is essential.
What a Certificate of Analysis Should Confirm
Quality research-grade DSIP should come with independent laboratory documentation showing:
- HPLC purity at or above 98 percent
- Mass spectrometry confirmation of the correct molecular weight (849.9 Da for Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu)
- Amino acid sequence verification of the full nine-amino-acid chain
- Batch number and accredited third-party laboratory certification
Our guide on How to Read a Peptide Certificate of Analysis explains each section of a peptide CoA document so you know exactly what to look for before purchasing.
Recommended Research Supplier
Peptides Finder connects researchers with vetted suppliers providing full third-party testing documentation for DSIP and all major neurological and sleep research peptides.
Browse DSIP Research Compounds at Peptides Finder
We may earn a commission from purchases made through links on this page.
Frequently Asked Questions
What is DSIP peptide and what does it do?
DSIP stands for delta sleep-inducing peptide. It is a naturally occurring nine-amino-acid neuropeptide first isolated in 1974 from the blood of sleeping rabbits by Swiss researchers at the University of Basel. DSIP promotes the deepest stage of sleep, slow-wave or delta sleep, through a multi-system mechanism that includes suppressing stress hormones (particularly ACTH and cortisol through the HPA axis), modulating GABAergic signaling in sleep-regulatory brain centers, influencing thalamic oscillation patterns toward slow-wave frequencies, and supporting the nighttime growth hormone pulse that occurs during deep sleep. It has also demonstrated pain-modulating and antioxidant properties in published research.
How was DSIP discovered?
DSIP was discovered in 1974 by Marcel Monnier and his team at the University of Basel in Switzerland through a cross-circulation experiment with rabbits. Two rabbits were surgically connected so their blood could flow between them. When the thalamus of one rabbit was electrically stimulated to produce deep sleep, blood from that sleeping rabbit was collected and injected into an awake rabbit. The awake rabbit entered the same pattern of deep delta sleep. After extensive biochemical analysis of the blood, Monnier's team isolated the nine-amino-acid peptide responsible for transferring the sleep state and named it the delta sleep-inducing peptide. This experiment was one of the first demonstrations that sleep could be regulated by a specific bloodborne chemical signal.
How does DSIP improve sleep quality?
DSIP improves sleep quality primarily by increasing the duration and depth of slow-wave (delta) sleep, which is the most physically and neurologically restorative stage of the sleep cycle. It accomplishes this through several pathways acting together: it suppresses ACTH and cortisol through HPA axis inhibition, removing the stress hormone barrier that prevents deep sleep; it enhances GABAergic inhibitory tone in sleep-promoting brain regions; and it modulates thalamic neural activity toward the slow oscillatory patterns that define delta sleep on EEG measurements. Research subjects show faster sleep onset, fewer nighttime awakenings, greater time in slow-wave sleep, and improved next-day subjective recovery ratings compared to placebo.
Does DSIP cause morning grogginess or next-day sedation?
No. This is one of DSIP's most clinically important distinctions from conventional sleep medications. Drugs like benzodiazepines and Z-drugs (zolpidem, eszopiclone) produce sleep partly through broad CNS sedation, and this sedation often lingers into the morning as grogginess, impaired coordination, and cognitive blunting. DSIP works by deepening and promoting natural sleep architecture rather than pharmacologically sedating the brain. The sleep it promotes is genuine slow-wave sleep with normal sleep-stage cycling, and subjects in published research consistently report normal or improved alertness and cognitive performance the following morning.
What is the typical research dosing for DSIP?
Published human-context DSIP research has most commonly used doses in the 200 to 500 mcg range administered subcutaneously or intravenously. Lower doses in the range of 25 to 50 mcg per kilogram of body weight have been used in animal studies for dose-response characterization. The most consistent clinical protocol is a single pre-sleep subcutaneous injection administered 30 to 60 minutes before intended sleep time. Some research has used 5 to 7 day consecutive courses, while other protocols use DSIP on specific nights rather than continuously. Published research does not document significant tolerance development, so both course-based and as-needed approaches appear viable depending on the study design.
Can DSIP be combined with other sleep or recovery peptides?
Yes, and some of the most interesting research combinations involve DSIP alongside compounds that address complementary aspects of sleep and recovery. Combining DSIP with epithalon addresses both the acute slow-wave sleep promotion (DSIP) and the longer-term restoration of the pineal-melatonin circadian system (epithalon). Combining DSIP with GHRPs like hexarelin or ipamorelin provides both the natural sleep-coupled GH release that DSIP's deep sleep promotion enables and a direct GH stimulation from the GHRP, potentially amplifying total nighttime GH output through two complementary pathways. Researchers combining DSIP with other GABAergic compounds should monitor for additive sedation effects.
Is DSIP safe for research use?
Published research spanning five decades has consistently found DSIP to be well-tolerated at standard research doses. No serious adverse events have been directly attributed to DSIP in the published literature. Common minor observations include brief post-injection warmth or flushing and mild injection site reactions, both self-resolving. Unlike benzodiazepines and opioid-based analgesics, DSIP does not produce physical dependence or significant withdrawal effects in published research. The main research safety consideration is potential additive sedation when combined with other CNS-depressant compounds, which should be accounted for in protocol design.
Conclusion: DSIP and the Science of Restorative Sleep Research
Sleep science has come a long way since Marcel Monnier injected sleeping rabbit blood into an awake rabbit in 1974. What began as a curious cross-circulation experiment has grown into five decades of research on one of the most biologically complex peptides in the body.
DSIP stands at the intersection of three of the most important areas of modern health science: sleep biology, stress physiology, and longevity research. The slow-wave sleep it promotes is the stage where growth hormone flows, where the brain cleans itself, where the immune system does its nightly maintenance, and where the body rebuilds from the day's demands. Losing this sleep stage is one of the most consequential ways the body ages. Restoring it is one of the most impactful interventions available for overall healthspan.
The fact that DSIP accomplishes this without sedation, without dependence risk, without morning grogginess, and through a mechanism that strengthens rather than overrides the body's natural sleep architecture makes it qualitatively different from any pharmacological sleep medication currently in clinical use.
Some questions about DSIP's exact mechanism remain open. The receptor pharmacology is not fully characterized. Human response is more variable than animal model data suggests. These are genuine scientific questions that ongoing research continues to address. But the reproducible effects on sleep architecture, stress hormones, and the downstream biological processes that deep sleep enables have been documented consistently enough across five decades to make DSIP one of the most scientifically credible tools in sleep research today.
Explore the full library of neurological, longevity, and recovery peptide research guides at Peptides Finder, and stay current with the latest developments in sleep science and neuropeptide research.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. DSIP is sold as a research chemical. Research peptides are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any research compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.