Selank: The Nootropic Peptide for Anxiety and Focus
Selank reduces anxiety and sharpens focus by modulating GABA and BDNF in the brain. The approved nootropic peptide's science, dosing, and research for 2026.
Anxiety and focus are two sides of the same neurological coin.
When your brain is running too hot with stress and fear signals, focused thinking becomes nearly impossible. The circuits that handle worry and the circuits that handle concentration compete for the same neural real estate. Most compounds that calm anxiety do it by sedating the brain. Benzodiazepines, for example, are effective at reducing anxiety but they also blunt alertness, impair memory formation, and carry a serious dependence risk.
Selank does something different. It reduces anxiety without sedation. It calms the fear circuits while simultaneously supporting the cognitive circuits that drive focus, memory, and mental clarity. Researchers describe this as an anxiolytic effect without the typical cognitive penalty that comes with conventional anxiety medications.
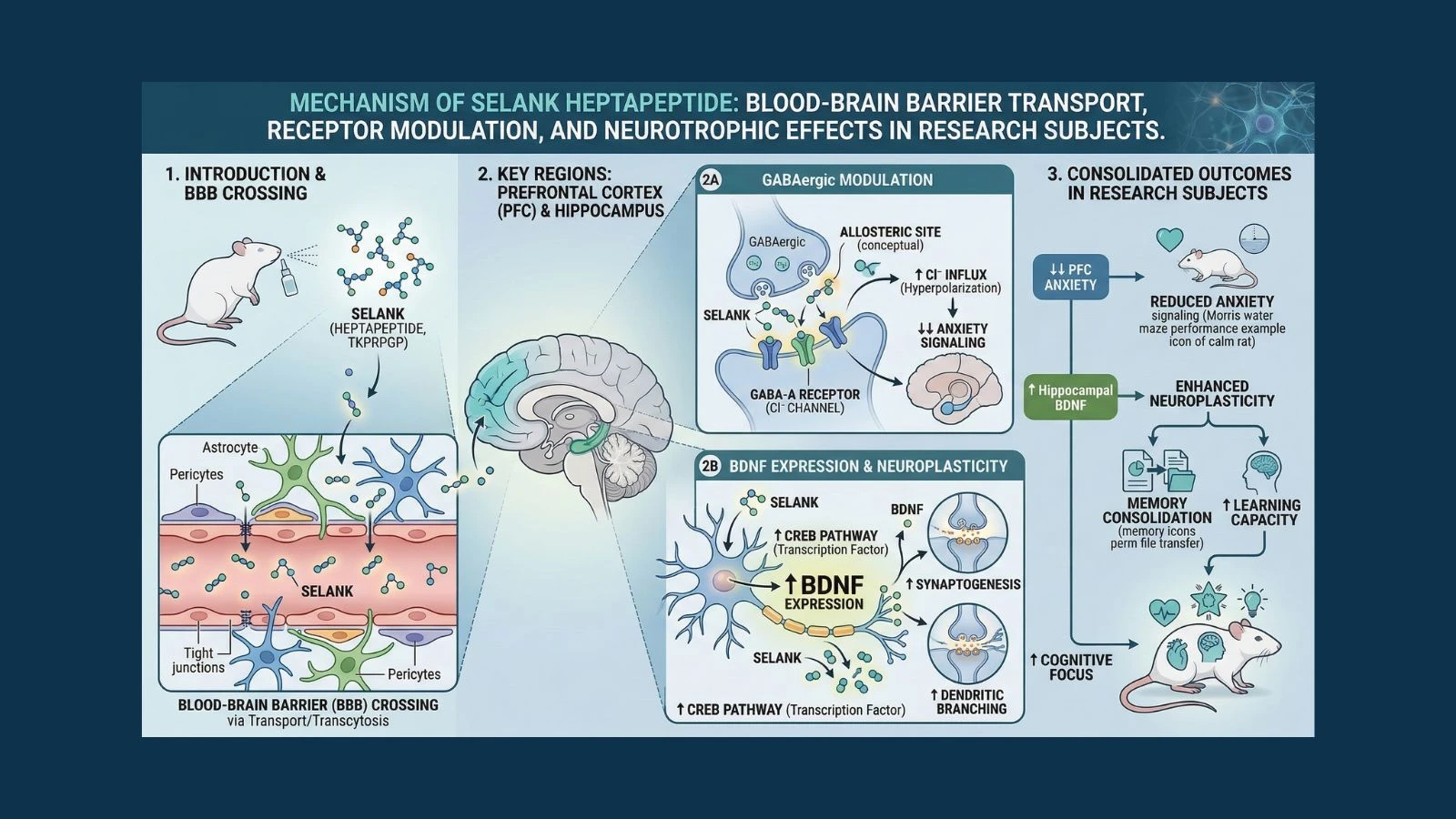
Selank is a synthetic heptapeptide (seven amino acids) developed in Russia that has been approved for medical use in Russia and Ukraine as an anxiolytic and nootropic agent. It has over three decades of research behind it, a favorable safety profile, and a mechanism of action that touches multiple brain systems simultaneously. For researchers studying anxiety neuroscience, cognitive enhancement, neuroplasticity, and stress biology, selank is one of the most scientifically interesting neuropeptides in current study.
To understand how neuropeptides broadly influence brain states, our guide on Neuropeptide S: The Science of Arousal, Anxiety, and Neurological Research provides essential foundational context before diving into selank's specific mechanisms.
What Is Selank? Origins and Structure
Selank was developed at the Institute of Molecular Genetics of the Russian Academy of Sciences by Professor Valery Seredenin and his team. It was created as a synthetic analog of a naturally occurring tetrapeptide called tuftsin, which is a fragment of the immunoglobulin IgG molecule.
Tuftsin itself has immune-modulating and some anxiolytic properties, but it is unstable in the body and breaks down too quickly to be useful as a research or clinical compound. Scientists modified tuftsin by extending it with three additional amino acids to create a seven-amino-acid sequence: Thr-Lys-Pro-Arg-Pro-Gly-Pro. This heptapeptide, named selank, is significantly more stable than tuftsin while preserving and amplifying its beneficial effects.
The addition of the extra amino acids also changed how selank interacts with the brain. While tuftsin primarily affects the immune system, selank shows stronger central nervous system activity, crossing the blood-brain barrier and directly influencing multiple neurotransmitter systems involved in anxiety, cognition, and stress response.
Regulatory Status
Selank holds an important distinction in the peptide research world: it is not just a research compound but an approved pharmaceutical medication in Russia, where it is prescribed under the name Selank as an anxiolytic and nootropic. It has also received approval in Ukraine. This regulatory status means it has undergone formal clinical trial evaluation, and its safety and efficacy data meet the standards of at least two national regulatory bodies, giving it considerably more clinical credibility than most research peptides.
In the United States and European Union, selank is classified as a research peptide and is not approved for human use, but the existing Russian clinical data provides a valuable foundation for ongoing research.
For broader context on the distinction between approved medications and research peptides, our guide on GLP-1 Agonists vs. Peptides: Medications vs. Research Compounds explains the regulatory landscape in detail.
How Selank Works: A Multi-System Brain Mechanism
Selank's effects on anxiety and cognition come from its ability to interact with multiple brain systems simultaneously. Unlike drugs that act on a single receptor type, selank modulates several interconnected pathways, which is thought to be why it produces a balanced calming and clarity-enhancing effect rather than the one-dimensional sedation of conventional anxiolytics.
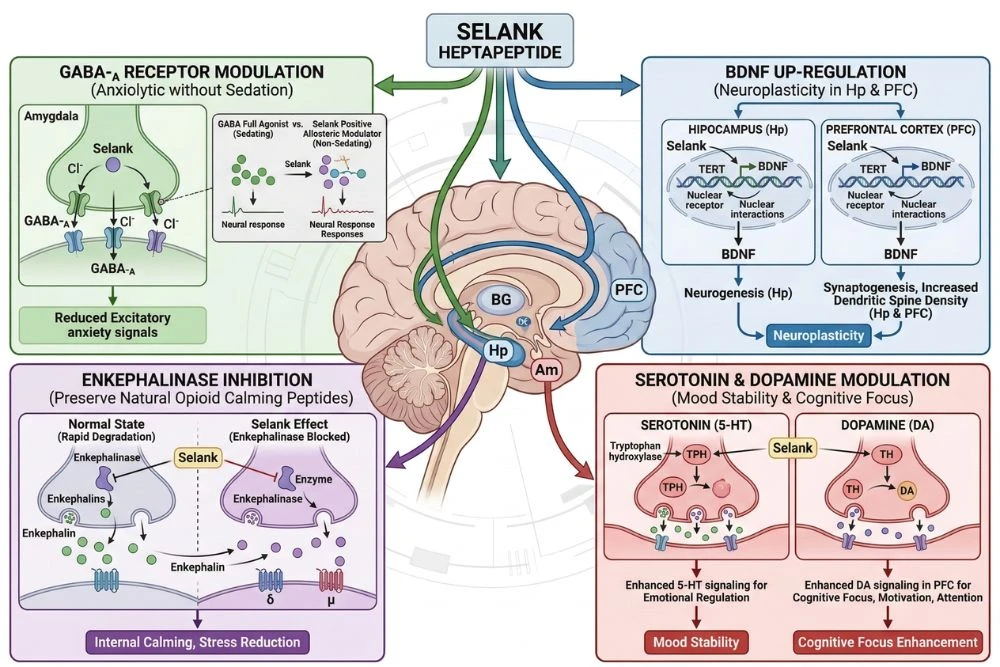
The GABA System: Calming Without Sedation
GABA (gamma-aminobutyric acid) is the brain's primary inhibitory neurotransmitter. When GABA binds to GABA-A receptors on neurons, it reduces their excitability, creating a calming effect throughout the nervous system. Benzodiazepines work by strongly potentiating GABA-A receptors, which is why they produce powerful sedation alongside their anxiety relief.
Selank modulates the GABA system, but with important differences. Research shows that selank enhances GABAergic tone in a more selective and gentler way than benzodiazepines, reducing anxiety-related neural excitability without causing the widespread CNS depression that produces sedation, memory impairment, and dependence associated with benzodiazepine use.
A study published in PMC confirmed that selank produces its anxiolytic effect through GABAergic mechanisms, with a profile of receptor interaction significantly different from classical benzodiazepines, explaining its anxiolytic effect without the cognitive impairment and sedation that typically accompany GABA-potentiating drugs.
BDNF Upregulation: The Neuroplasticity Connection
One of selank's most important and distinctive mechanisms is its ability to increase the expression of BDNF (brain-derived neurotrophic factor). BDNF is often called the brain's fertilizer. It promotes the growth of new neurons, strengthens synaptic connections, and is essential for learning, memory formation, and cognitive resilience.
BDNF levels are reduced by chronic stress, anxiety disorders, and depression. This reduction is one of the key reasons chronic stress impairs memory and learning over time. Selank's ability to upregulate BDNF is significant because it means the peptide is not just calming the brain in the moment but actively supporting the neural architecture needed for better thinking over the longer term.
Research published in PubMed demonstrated that selank significantly increased BDNF mRNA expression in rat hippocampal tissue and prefrontal cortex, two brain regions critical for memory consolidation and executive function, providing a molecular basis for selank's observed cognitive enhancement effects alongside its anxiolytic properties.
Enkephalin Breakdown Inhibition
Enkephalins are natural opioid peptides produced in the brain that are involved in mood regulation, stress response, and pain modulation. They bind to opioid receptors and produce feelings of calm, wellbeing, and emotional stability. The problem is that enkephalins are broken down very rapidly by enzymes called enkephalinases.
Selank has been shown to inhibit enkephalinase activity, slowing the breakdown of natural enkephalins and extending their presence in the brain. This mechanism contributes to selank's mood-stabilizing and anxiolytic effects through a pathway completely separate from its GABAergic activity. By preserving the brain's own natural calming peptides, selank amplifies the endogenous stress-buffering system.
Serotonin and Dopamine Modulation
Research has also documented selank's influence on monoamine neurotransmitter systems. Selank modulates serotonin metabolism in the brain, which contributes to its anxiolytic and mood-stabilizing properties. Serotonin is closely associated with feelings of wellbeing, emotional regulation, and anxiety control.
Additionally, selank appears to influence dopamine signaling in areas associated with motivation and cognitive performance. This dopaminergic component is thought to contribute to the focus and mental energy enhancement that research subjects report alongside the reduction in anxiety. It is this combination of serotonin stabilization for emotional calm and dopamine support for cognitive drive that gives selank its unique dual character as both an anxiolytic and a nootropic.
The Blood-Brain Barrier and Bioavailability
A key practical characteristic of selank is its ability to cross the blood-brain barrier efficiently, particularly via intranasal administration. The intranasal route delivers the peptide directly to the olfactory nerve endings, which provide a direct pathway to the brain bypassing the systemic circulation. This is why the intranasal nasal spray formulation is the primary clinical delivery method in Russia rather than injection.
This intranasal bioavailability is one of selank's practical research advantages over peptides that require injection for central nervous system effects, making it more accessible for study designs where injection compliance or comfort is a variable.
The Research Benefits of Selank
Three decades of Russian research, combined with growing international interest, have produced a well-characterized picture of selank's biological effects. Here is what the evidence shows across key research areas.
Benefit 1: Anxiety Reduction Without Cognitive Impairment
This is selank's defining research achievement and the reason it attracted serious clinical interest in Russia. In multiple studies comparing selank to classical benzodiazepines, selank consistently matched or exceeded the anxiolytic effect of benzodiazepines on standardized anxiety measures, while producing none of the cognitive side effects including sedation, memory impairment, or psychomotor slowing.
A clinical study published in PubMed compared selank to medazepam (a classic benzodiazepine) in patients with generalized anxiety disorder and found that both compounds produced comparable reductions in anxiety symptoms. However, only selank showed simultaneous improvements in cognitive performance metrics including attention, memory, and mental processing speed. The benzodiazepine group showed the expected cognitive decline alongside anxiety relief.
For research into treatments for anxiety disorders, this combination of full anxiolytic efficacy with intact and even improved cognition represents a significant scientific advance over existing pharmacological approaches.
Benefit 2: Cognitive Enhancement and Nootropic Effects
Selank's nootropic classification comes from consistent research findings showing improvements in several cognitive domains, particularly under conditions of stress or cognitive load:
- Attention and concentration: Research subjects show improved sustained attention and reduced distractibility on standardized cognitive tasks
- Memory consolidation: BDNF upregulation in the hippocampus directly supports the formation and consolidation of new memories, with research showing improved recall performance
- Mental processing speed: Subjects demonstrate faster information processing and decision-making responses under stressful or complex task conditions
- Working memory: Improvements in holding and manipulating information in short-term memory, which is critical for complex reasoning and problem-solving
These cognitive improvements are thought to result from the combination of reduced anxiety-related interference with cognitive processing and the direct neuroplasticity support provided by BDNF upregulation.
Benefit 3: Stress Resilience and Adaptogenic Properties
Selank demonstrates what researchers describe as adaptogenic properties, meaning it helps the brain maintain homeostasis under various stressors rather than simply suppressing a single anxiety pathway. When research subjects are placed under cognitive or emotional stress conditions, selank-treated groups show more stable physiological and psychological responses compared to controls.
This adaptogenic quality is linked to selank's multi-system mechanism. Because it supports the GABA, enkephalin, serotonin, and BDNF systems simultaneously, the brain has broader resilience against stress from multiple angles rather than just one neurotransmitter system being boosted while others remain vulnerable.
Benefit 4: Immune System Modulation
As a tuftsin analog, selank retains meaningful immune-modulating activity alongside its central nervous system effects. Research has documented selank's ability to modulate the expression of interleukins and other immune signaling molecules, influencing the balance between pro-inflammatory and anti-inflammatory immune activity.
The connection between immune function and mental health is well-established through the field of psychoneuroimmunology. Chronic inflammation is associated with depression and anxiety, and compounds that simultaneously address both neural and immune dysregulation may have synergistic effects on mental health outcomes. Selank's dual neural and immune activity makes it a uniquely interesting compound for research at this intersection.
For a dedicated look at how peptides influence immune function and the mind-body connection, our guide on Thymosin Alpha 1: The Gold Standard for Immune Optimization explores the immune-peptide relationship in depth.
Benefit 5: Opioid Withdrawal and Substance Research
An emerging and compelling research application for selank is its potential role in managing anxiety and dysphoria associated with opioid and alcohol withdrawal. Because selank supports the enkephalin system (the brain's natural opioid signaling) and stabilizes GABAergic tone, it addresses two of the primary neurological mechanisms underlying withdrawal anxiety and craving.
Animal models of opioid and alcohol dependence have shown that selank reduces withdrawal-associated anxiety behaviors and helps normalize neurotransmitter function during the post-dependence recovery period. This positions selank as an interesting research tool for addiction medicine and withdrawal management science.
Selank vs. Conventional Anxiety Treatments: A Research Comparison
Understanding selank's place in the anxiety treatment research landscape requires comparing it directly to the primary existing pharmacological approaches.
Selank vs. Benzodiazepines
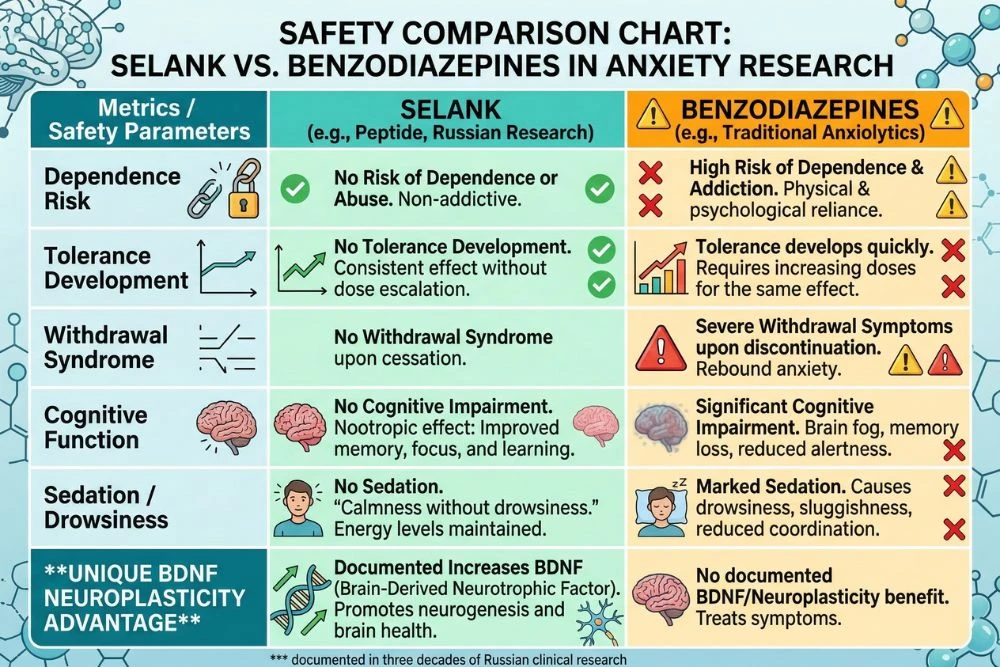
Benzodiazepines (diazepam, alprazolam, clonazepam) are the gold standard for acute anxiety relief. They work by strongly potentiating GABA-A receptors, producing rapid and powerful sedation and anxiety reduction. Their limitations are well-documented: cognitive impairment, memory suppression, psychomotor slowing, tolerance with chronic use, physical dependence, and significant withdrawal syndrome.
Selank produces comparable anxiolytic effects through a more selective GABAergic modulation that does not carry the cognitive penalty, tolerance, or dependence profile. It does not bind to the benzodiazepine site on GABA-A receptors, which is believed to be why it avoids the sedation and dependence risks. For research into safer long-term anxiety management, selank represents a fundamentally different mechanistic approach.
Selank vs. SSRIs
Selective serotonin reuptake inhibitors (SSRIs) are the most widely prescribed medications for anxiety disorders and depression. They work by blocking serotonin reuptake, increasing serotonin availability in synapses. SSRIs require 2 to 6 weeks of daily use before producing therapeutic effects and are associated with a range of side effects including initial anxiety exacerbation, sexual dysfunction, weight changes, and emotional blunting.
Selank produces effects within hours to days rather than weeks, does not require daily ongoing use for benefit, and does not carry the same side effect profile. For research into rapid-onset anxiety interventions, selank's speed of action is a significant practical advantage. It does not replace the chronic serotonin modulation that SSRIs provide for severe depression, but for anxiety-focused research, the faster and more flexible profile is clinically relevant.
Selank vs. Semax (Related Nootropic Peptide)
Semax is another Russian-developed neuropeptide that is frequently discussed alongside selank. Both are approved in Russia, both increase BDNF, and both improve cognitive performance. The key difference is in their primary emotional effect: semax is primarily stimulating and energizing, increasing alertness and drive, while selank is primarily calming and anxiety-reducing while still supporting cognition.
Researchers studying neurological enhancement often consider selank and semax as complementary compounds. Selank is preferred when anxiety, stress sensitivity, or emotional dysregulation is the primary research target. Semax is preferred when energy, motivation, and cognitive activation are the primary targets. When anxiety and cognitive enhancement are both goals simultaneously, selank is typically the starting point.
Our upcoming guide on Semax: The Nootropic Peptide for Focus, Memory, and Brain Health will cover semax's distinct mechanism and applications in detail for researchers interested in the full Russian neuropeptide research program.

Research Protocols and Dosing Considerations
Important note: The following information is drawn from published research studies and the clinical use data from Russia and Ukraine where selank is an approved medication. Selank is not approved by the FDA for human use. This section is for educational and scientific reference only.
Administration Routes
Selank is unique among research peptides in that its primary clinical administration route is intranasal rather than subcutaneous injection. The intranasal route leverages the olfactory pathway to deliver selank directly to the brain, bypassing first-pass metabolism and the blood-brain barrier challenge that makes many larger peptides difficult to administer without injection.
In Russian clinical practice, selank is supplied as a 0.15 percent nasal spray solution. Research protocols use both intranasal and subcutaneous injection routes depending on the study design, with intranasal being the more practical option for human research and subcutaneous being used in animal models or when precise systemic dosing is required.
Intranasal Dosing in Research
In the Russian clinical approval framework, the standard protocol for selank nasal spray is:
- Concentration: 0.15 percent solution (1.5 mg per mL)
- Dose per administration: 2 to 3 sprays per nostril (approximately 250 to 400 mcg per administration)
- Frequency: 2 to 3 times daily for acute anxiety management
- Course duration: Typically 10 to 14 days for a standard treatment course
Subcutaneous Research Dosing
For injectable research protocols, selank has been studied at doses ranging from 200 to 500 mcg per administration, with most research centering on the 250 to 300 mcg range for human-scale studies. In animal models, dosing is typically adjusted for body weight.
The onset of effects with subcutaneous administration is typically within 20 to 40 minutes, similar to intranasal delivery. The duration of noticeable cognitive and anxiolytic effects typically lasts 4 to 8 hours per administration in published research.
Cycling and Duration
Unlike some research peptides that require careful cycling to prevent desensitization, selank does not appear to cause receptor downregulation or tolerance with repeated use in published research. The Russian clinical protocols use it in defined courses (typically 10 to 14 days) followed by breaks, which appears to be driven more by clinical convention and the goal of course-based treatment evaluation than by pharmacological necessity.
Some research subjects have used selank on an as-needed basis for acute anxiety situations rather than in defined courses, and published data does not indicate any loss of efficacy with this approach.
Reconstitution for Research Use
For research settings where selank is obtained as lyophilized powder, it is reconstituted with either bacteriostatic water for injection or sterile saline. For intranasal preparation, reconstitution targets a dilute solution appropriate for nasal spray delivery.
Our Peptide Calculator Guide provides the step-by-step process for reconstitution and precise dosing calculations for any research peptide including selank.
Safety Profile: What Three Decades of Research Shows
Selank's safety profile is one of its most compelling research characteristics. Three decades of study in Russia, combined with clinical approval and ongoing use as an approved medication, have produced a well-characterized tolerability picture.
Observed Side Effects
Selank is considered one of the best-tolerated research neuropeptides. In clinical studies and reported clinical use, the most commonly noted effects are:
- Mild nasal irritation with intranasal administration, typically transient and well-tolerated
- A brief initial sensation of warmth or mild dizziness in some subjects shortly after administration, resolving within 15 to 30 minutes
- In a small subset of subjects, mild drowsiness in the first 1 to 2 hours after initial doses, which typically diminishes with continued use
No serious adverse events have been reported in published clinical research at standard dosing ranges. Unlike benzodiazepines, selank shows no evidence of physical dependence, withdrawal syndrome, or tolerance development in the published literature.
No Dependence or Withdrawal
This is perhaps the most clinically significant safety characteristic of selank compared to conventional anxiolytics. Benzodiazepine dependence and withdrawal are among the most challenging pharmacological problems in modern psychiatry, with withdrawal symptoms that can include severe rebound anxiety, seizures, and protracted neurological disruption.
Selank's mechanism does not involve the same receptor binding sites that produce benzodiazepine tolerance and dependence. Published research and decades of clinical use in Russia have not demonstrated any evidence of physical dependence or significant withdrawal effects when selank courses are discontinued, even after extended periods of use.
Interaction Considerations
Because selank modulates multiple neurotransmitter systems, researchers designing protocols that combine selank with other neuroactive compounds should consider potential additive effects on GABAergic or serotonergic systems. Combining selank with other GABA-potentiating compounds may produce stronger sedation than either compound alone. Selank does not appear to interact significantly with standard non-psychoactive research peptides operating outside the central nervous system.
The Neuroscience of Anxiety and How Selank Addresses It
To fully appreciate what selank offers as a research tool, it helps to understand what anxiety actually is at the brain level and why it is so hard to treat without cognitive side effects.
The Amygdala and the Fear Response
Anxiety at its core is a hyperactive fear response. The amygdala, an almond-shaped structure deep in the brain's limbic system, is the primary fear-processing center. It monitors the environment for threats and triggers the fight-or-flight stress response when it detects danger. In anxiety disorders, the amygdala is chronically overactivated, interpreting non-threatening situations as dangerous and generating persistent fear and stress responses.
The prefrontal cortex (PFC) is supposed to regulate amygdala activity, providing rational perspective that damps down unnecessary fear responses. In anxiety disorders, this top-down regulation by the PFC is weakened, allowing the amygdala to run hot without adequate brakes.
Selank addresses this imbalance from multiple directions: its GABAergic modulation reduces overall neural excitability including in the amygdala and its associated circuits, while its BDNF-enhancing effects support the health and connectivity of PFC circuits that regulate fear, strengthening the top-down control that anxiety disorders undermine.
Why Focus Improves When Anxiety Decreases
The cognitive impairment that comes with anxiety is not a separate problem from the anxiety itself. It is caused by it. When the amygdala and stress circuits are highly active, they compete for processing resources with the prefrontal and hippocampal circuits that handle focused thinking and memory formation. Cortisol and other stress hormones actively impair PFC function when chronically elevated.
This is why simply reducing anxiety, when done correctly without cognitive side effects, automatically improves focus. Selank's approach of calming fear circuits while simultaneously supporting cognitive circuits with BDNF produces a synergistic benefit: anxiety relief clears the interference while the nootropic effects directly enhance the cognitive signal. The result is a clearer, more focused mental state than either effect alone would produce.
BDNF and Long-Term Brain Health
The BDNF upregulation that selank produces has implications beyond the acute research session. BDNF is critical for neurogenesis (the birth of new neurons, particularly in the hippocampus), long-term potentiation (the cellular mechanism of learning and memory), and the maintenance of existing neural connections. Chronic stress, anxiety, and depression all reduce BDNF, contributing to the shrinkage of hippocampal volume that is observed in long-term anxiety disorder patients.
Selank's ability to restore and elevate BDNF means it is potentially doing more than managing anxiety symptoms. It may be supporting the structural repair and maintenance of brain circuits that anxiety disorders progressively degrade. This neuroplastic dimension of selank's research profile is one of the most forward-looking aspects of its scientific story.
Sourcing Quality Selank for Research
Selank is a heptapeptide with a defined and well-documented amino acid sequence, making quality verification through standard analytical methods straightforward. As with all research peptides, third-party testing is essential.
What a Certificate of Analysis Should Confirm
Research-grade selank should come with independent laboratory documentation showing:
- HPLC purity at or above 98 percent
- Mass spectrometry confirmation of the correct molecular weight (751.86 Da for the Thr-Lys-Pro-Arg-Pro-Gly-Pro sequence)
- Amino acid sequence verification
- Batch number and accredited third-party laboratory certification
For a plain-language guide to reading and evaluating peptide CoA documentation, our How to Read a Peptide Certificate of Analysis covers every section you need to verify.
Intranasal vs. Injectable Sourcing
Researchers should verify whether they are sourcing selank intended for reconstitution as an injectable solution or as an intranasal preparation. The concentration, carrier solutions, and buffering agents differ between these formats. Some suppliers provide selank in nasal spray solution format already prepared, while others provide lyophilized powder for the researcher to reconstitute in the appropriate vehicle for their protocol.
Recommended Research Supplier
Peptides Finder connects researchers with vetted suppliers who provide full third-party testing documentation for selank and all major nootropic and neurological research peptides.
Browse Selank Research Compounds at Peptides Finder
We may earn a commission from purchases made through links on this page.
Frequently Asked Questions
What is selank and how does it work?
Selank is a synthetic heptapeptide (seven amino acids) developed at the Russian Academy of Sciences as a stable analog of the natural immune peptide tuftsin. It works through multiple brain systems simultaneously: it modulates GABA-A receptors to reduce anxiety without sedation, increases BDNF expression to support neuroplasticity and cognitive function, inhibits the enzyme that breaks down natural enkephalins (the brain's own calming opioid peptides), and modulates serotonin and dopamine signaling for mood and focus. This multi-system approach is what gives selank its unique profile of anxiety relief combined with cognitive enhancement rather than the cognitive impairment that conventional anxiolytics typically cause.
Is selank approved for human use?
Yes, in Russia and Ukraine. Selank is an approved pharmaceutical medication in both countries, where it is prescribed as an anxiolytic and nootropic agent for anxiety disorders, stress, and cognitive impairment. In the United States and European Union, selank is classified as a research peptide and is not approved by the FDA or EMA for human use. However, its regulatory approval in Russia means it has undergone formal clinical trial evaluation and its safety and efficacy data meet the standards of national regulatory authorities, giving it considerably more clinical credibility than most research compounds that lack any regulatory approval history.
How does selank compare to benzodiazepines for anxiety?
Clinical research comparing selank to benzodiazepines has shown comparable anxiolytic efficacy, meaning both reduce anxiety to a similar degree on standardized measures. The critical difference is the side effect profile. Benzodiazepines impair cognitive function, cause sedation, carry a risk of physical dependence, and produce a significant withdrawal syndrome when discontinued. Selank produces anxiety relief without cognitive impairment, in fact simultaneously improving cognitive performance in research settings, and does not demonstrate tolerance, dependence, or withdrawal in published research. For researchers studying anxiety treatment, selank represents a fundamentally different mechanistic approach with a significantly more favorable tolerability profile.
What cognitive benefits does selank produce?
Research has documented selank-associated improvements in several cognitive domains including sustained attention and concentration, working memory, mental processing speed under cognitive load, and memory consolidation. These effects are thought to result from two complementary mechanisms: the reduction of anxiety-related interference with cognitive processing (since anxiety actively impairs PFC function and competes with cognitive resources) and the direct neuroplasticity support provided by selank's upregulation of BDNF in the hippocampus and prefrontal cortex. BDNF is essential for synaptic strengthening, memory formation, and the maintenance of neural circuit integrity.
How is selank administered in research?
Selank is unique among research neuropeptides in that its primary clinical administration route is intranasal rather than subcutaneous injection. The intranasal route delivers selank directly to brain tissue via the olfactory pathway, bypassing the systemic circulation and the blood-brain barrier challenge. In the Russian clinical approval framework, the standard protocol uses a 0.15 percent nasal spray solution administered 2 to 3 times daily in 10 to 14-day courses. Injectable subcutaneous administration is also used in research settings, particularly in animal models or protocols requiring precise systemic dosing, typically at 250 to 300 mcg per administration.
Does selank cause dependence or withdrawal?
Published research and decades of clinical use in Russia have not demonstrated physical dependence or significant withdrawal effects from selank, even after extended use. This contrasts sharply with benzodiazepines, where physical dependence and withdrawal are among the most serious clinical challenges. Selank does not bind to the benzodiazepine binding site on GABA-A receptors, which is thought to be the primary reason it avoids the tolerance and dependence mechanisms that make benzodiazepines problematic for long-term use. This absence of dependence risk is one of selank's most clinically meaningful safety advantages.
Can selank be used alongside other research peptides?
Selank's primary mechanism is in the central nervous system, while many other research peptides operate primarily on peripheral systems (muscle tissue, the hormonal axis, connective tissue repair). This means selank generally does not interact pharmacologically with non-neuroactive research peptides. However, researchers combining selank with other compounds that affect the central nervous system, including other GABAergic agents, serotonergic compounds, or opioid-related peptides, should account for potential additive effects. In Russian clinical research, selank has been studied as part of comprehensive neurological research protocols, and its immune-modulating properties make it potentially complementary to immune-focused peptides like thymosin alpha 1 in protocols addressing the psychoneuroimmunological dimension of health.
Conclusion: Selank as the Benchmark for Nootropic Anxiety Research
The challenge of treating anxiety without impairing cognition is one of the oldest unsolved problems in neuropharmacology. Conventional solutions have always involved a trade-off: reduce the anxiety, lose the mental edge. Selank breaks that trade-off.
By working through multiple complementary brain systems simultaneously, selank produces anxiety relief through its GABAergic and enkephalinase-inhibiting effects while simultaneously enhancing the cognitive architecture that anxiety disorders erode. The BDNF upregulation it drives is not a side effect. It is part of the same integrated mechanism that makes selank qualitatively different from any conventional anxiolytic in the research toolkit.
Three decades of Russian research have given selank something rare in the peptide world: real clinical data, regulatory approval in multiple countries, and a long safety record with a therapy-scale human population. For researchers studying anxiety neuroscience, nootropic enhancement, stress biology, or the intersection of immune and neurological function, selank is not just worth understanding. It is essential.
The fact that it can be administered intranasally, produces effects within hours rather than weeks, carries no dependence risk, and improves rather than impairs cognition makes it one of the most practically useful neurological research tools in the current peptide landscape.
Explore the complete library of nootropic, neurological, and longevity peptide research guides at Peptides Finder, and stay current with the latest developments in neuropeptide science.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. Selank is approved as a pharmaceutical medication in Russia and Ukraine and is available as a research chemical in other jurisdictions. Outside of countries where it holds regulatory approval, selank is intended strictly for laboratory research and is not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any research compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.