MOTS-c: The Mitochondrial Peptide for Endurance
Discover how MOTS-c, a mitochondria-encoded peptide, boosts endurance, activates AMPK, improves insulin sensitivity, and may slow age-related metabolic decline.
For decades, scientists believed they had a complete picture of what mitochondria do. These tiny organelles inside each of your cells produce energy, regulate metabolism, and keep the lights on for every biological process in your body. Their genetic code was considered fully mapped, with 37 known genes and nothing left to discover. That assumption turned out to be wrong.
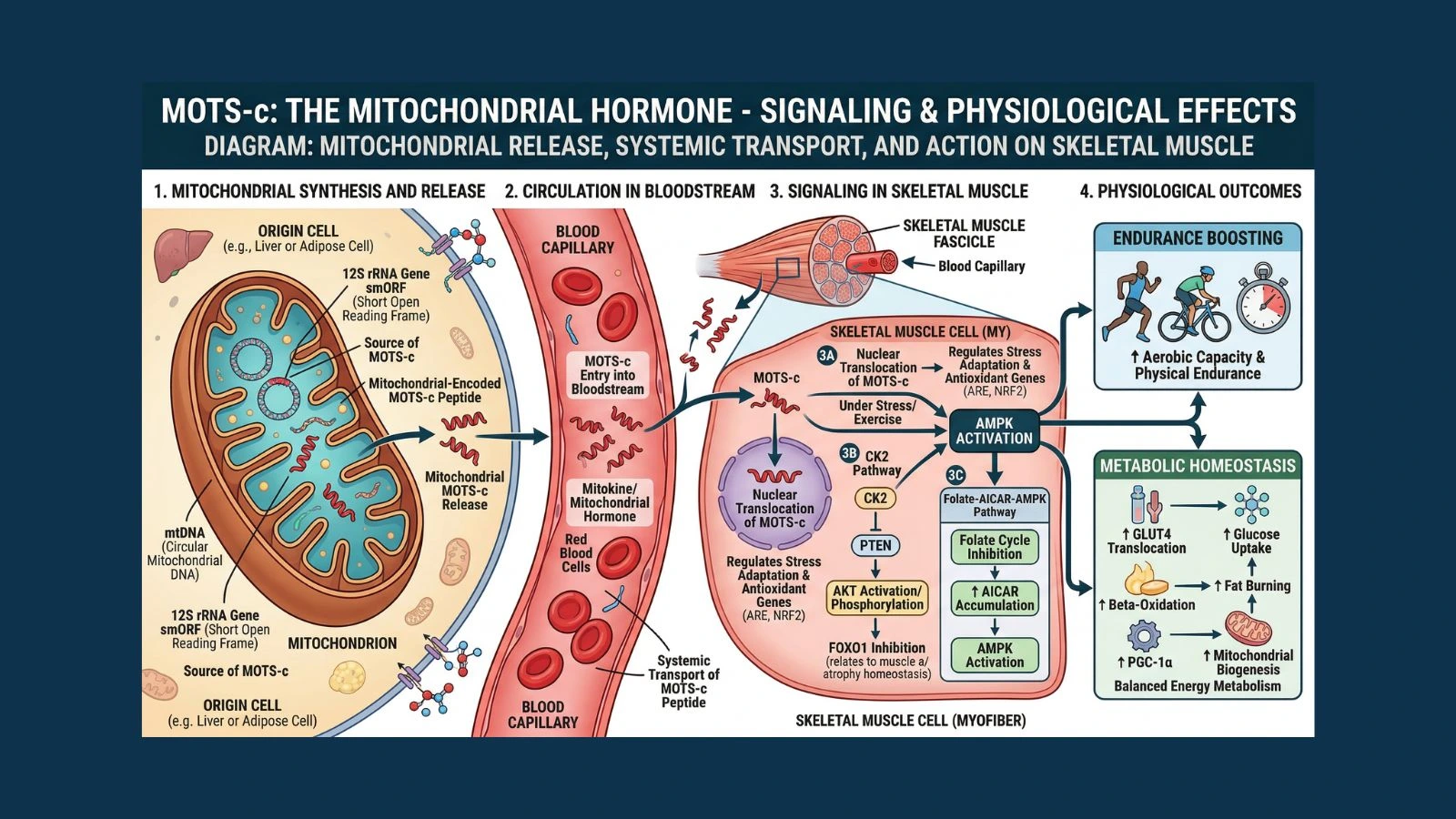
In 2015, researchers made a stunning find buried inside the mitochondrial genome: a hidden peptide encoded in a region that had been dismissed as non-coding. That peptide is MOTS-c. Short for Mitochondrial Open Reading Frame of the 12S rRNA-c, it is a 16-amino-acid signaling molecule that travels from mitochondria to skeletal muscle, activates a master metabolic switch inside cells, and produces effects that look remarkably similar to what happens when you exercise. That is why researchers have called it an "exercise mimetic," a compound that tells your body to perform many of the same metabolic changes that physical training triggers.
This guide covers what MOTS-c is, how it works at the molecular level, what the current research says about its effects on endurance and aging, and what researchers studying this peptide need to know before working with it.
What Is MOTS-c? A Peptide from an Unexpected Place
Most peptides and hormones that regulate metabolism are encoded by the nuclear genome, which is the DNA stored inside the cell's nucleus. MOTS-c is different. It is encoded directly in mitochondrial DNA (mtDNA), making it part of a new class of molecules called mitochondrial-derived peptides, or MDPs.
The mitochondrial genome is ancient, compact, and separate from nuclear DNA. Scientists long believed it only produced 13 proteins, all of which are components of the cell's energy-generating machinery. The discovery of MOTS-c, published in the landmark 2015 paper by Dr. Changhan Lee and colleagues at the University of Southern California, rewrote that understanding.
That discovery paper, published in Cell Metabolism, showed that MOTS-c is encoded in a short open reading frame within the 12S ribosomal RNA region of mtDNA. This is the same region that uses a mitochondrial-specific genetic code, which is why the peptide went undetected for so long. When the research team decoded it correctly, they found a biologically active peptide that circulates in human blood, is present in multiple tissues, and has meaningful effects on glucose metabolism and insulin sensitivity.
Since that initial discovery, more than 130 research articles have examined MOTS-c from different angles. This is exceptional velocity for a peptide less than a decade old, and it reflects how significant the discovery was. The idea that mitochondria produce hormones that regulate whole-body metabolism is still reshaping how biologists think about cellular signaling.
How MOTS-c Works: The Folate-AICAR-AMPK Pathway
Understanding MOTS-c means understanding one specific metabolic chain of events. When MOTS-c enters a cell, it targets the folate cycle. The folate cycle is a biochemical pathway that uses folate (a B vitamin) to process one-carbon units, which feed into DNA synthesis, amino acid production, and other critical processes.
MOTS-c inhibits key enzymes in this cycle. When the folate cycle slows down, the de novo purine biosynthesis pathway that runs in parallel with it also gets disrupted. This disruption causes a buildup of an intermediate molecule called AICAR (5-aminoimidazole-4-carboxamide ribonucleotide).
AICAR is a naturally occurring activator of AMPK, which stands for AMP-activated protein kinase. AMPK is one of the most important metabolic regulators in the human body. It is sometimes called the cell's "energy sensor" because it responds to low energy states and switches the cell into an energy-conserving, fuel-burning mode. When AMPK turns on, it:
- Promotes glucose uptake by muscle cells
- Increases fatty acid oxidation (fat burning for fuel)
- Stimulates mitochondrial biogenesis, meaning the creation of new mitochondria
- Activates PGC-1alpha, a key regulator of mitochondrial function and endurance adaptation
- Shifts the cell away from anabolic (building) processes and toward catabolic (energy-releasing) ones
This is the same pathway activated by metformin, the most widely prescribed diabetes drug in the world. The parallel between MOTS-c and metformin has attracted significant scientific attention, as outlined in the PMC full text of the 2015 discovery study. Both act on similar downstream targets, but MOTS-c does so as an endogenous peptide your own mitochondria produce rather than an external pharmaceutical compound.
A 2021 study in Biochimica et Biophysica Acta, confirmed by PubMed 33722744, demonstrated that MOTS-c and exercise work synergistically through this same AMPK/PGC-1alpha pathway. The study found that treadmill training elevated MOTS-c protein levels in skeletal muscle alongside AMPK phosphorylation and GLUT4 expression, providing direct molecular evidence for how MOTS-c and physical training reinforce each other.
MOTS-c and Endurance: What the Research Actually Shows
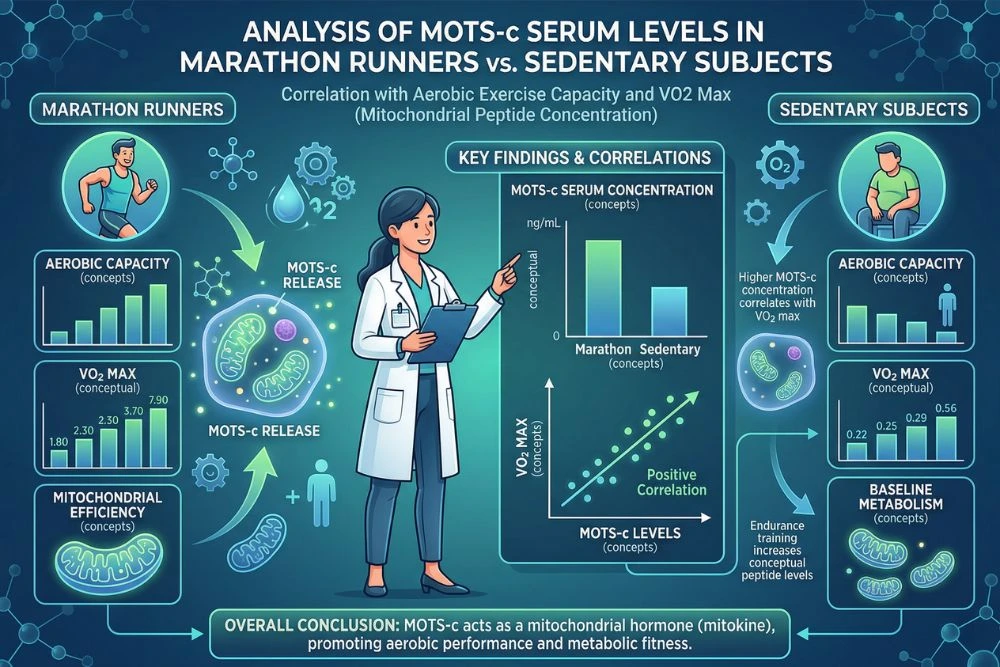
The connection between MOTS-c and physical endurance is one of the most exciting areas of active research. Evidence comes from multiple directions: studies measuring how exercise affects MOTS-c levels, animal studies where MOTS-c is administered directly, and human observational studies comparing athletes to sedentary individuals.
MOTS-c Rises During Exercise
One of the clearest findings in MOTS-c research is that physical exercise increases circulating MOTS-c levels. A 2025 study published in PubMed (PMID 39706498) compared serum MOTS-c concentrations in 10 experienced marathon runners against 12 sedentary control subjects. The results showed a significant positive correlation between MOTS-c levels and maximal oxygen uptake (VO2max), the gold standard measure of aerobic capacity. Endurance-trained athletes had meaningfully higher MOTS-c levels, and circulating MOTS-c correlated with lower body fat percentage and better aerobic performance metrics.
Importantly, the same research team then used a mouse model to investigate whether this relationship was causal. Mice subjected to eight weeks of endurance training showed increased plasma MOTS-c, improved mitochondrial respiratory function in skeletal muscle, and upregulated AMPK and PGC-1alpha expression in muscle tissue. This moves the connection beyond correlation and suggests that endurance training promotes MOTS-c secretion, which in turn enhances the mitochondrial machinery that supports further aerobic capacity.
Exogenous MOTS-c Mimics Exercise Effects
Perhaps the most remarkable finding in MOTS-c research comes from a 2021 study published in Nature Communications by Reynolds and colleagues at the University of Southern California. The team tested MOTS-c administration in mice across three age groups: young (2 months), middle-aged (12 months), and old (22 months).
Across all age groups, MOTS-c treatment significantly improved treadmill running performance. In old mice, the effect was especially striking. Compared to untreated controls, old mice receiving MOTS-c ran significantly farther and for longer on treadmill testing. The treatment also improved skeletal muscle metabolism, regulated nuclear gene expression including genes related to proteostasis, and helped myoblasts (muscle stem cells) adapt better to metabolic stress.
The study also showed that late-life MOTS-c administration, started at 23.5 months in mice (a very advanced age equivalent), was still effective. Even when treatment began late, MOTS-c increased physical capacity and markers of healthspan. This has important implications for research into aging and exercise decline.
MOTS-c Levels Decline with Age
Circulating MOTS-c is not static throughout life. Research shows that plasma MOTS-c concentrations decline with age, and this decline tracks closely with the metabolic dysfunction and muscle loss that characterize normal aging. Young, healthy individuals tend to have higher baseline MOTS-c, while older individuals show lower levels, particularly those with insulin resistance or metabolic disease.
This age-related decline creates a logical research question: if falling MOTS-c contributes to metabolic aging, can restoring MOTS-c levels reverse or slow that process? The animal data from the Reynolds 2021 study suggests yes, at least in terms of physical performance and muscle homeostasis. Human trials have not yet confirmed this, but the biological rationale is solid and active investigation is ongoing.
MOTS-c and Insulin Sensitivity: A Metabolic Optimizer
Insulin sensitivity refers to how efficiently your cells respond to insulin signals to absorb glucose from the bloodstream. When insulin sensitivity falls, as it does in type 2 diabetes and many cases of obesity, blood sugar stays elevated and metabolic disease follows. Restoring insulin sensitivity is one of the most important targets in metabolic medicine.
MOTS-c's original description in the 2015 Cell Metabolism paper was specifically framed around its ability to regulate insulin sensitivity. The discovery team found that:
- MOTS-c treatment in mice significantly increased glucose clearance during glucose tolerance tests
- Insulin-stimulated glucose disposal improved in MOTS-c-treated animals measured by clamp testing
- Mice fed a high-fat diet that received MOTS-c did not develop diet-induced obesity or insulin resistance, even though their calorie intake remained the same
- Age-dependent insulin resistance, which normally develops in older mice, was prevented by MOTS-c treatment
The mechanism runs through GLUT4, the glucose transporter protein that moves glucose into muscle cells. AMPK activation by MOTS-c promotes GLUT4 translocation to the cell membrane, meaning more glucose enters muscle tissue and less remains in the bloodstream. This is exactly the process that breaks down in type 2 diabetes, and it is the same mechanism improved by both exercise and metformin.
For researchers interested in metabolic disease, MOTS-c represents an endogenous signal that the body uses to regulate this process naturally. Understanding how that signal works, when it fails, and whether it can be restored is a major focus of current research.
MOTS-c as a Nuclear Regulator: Beyond the Cytoplasm
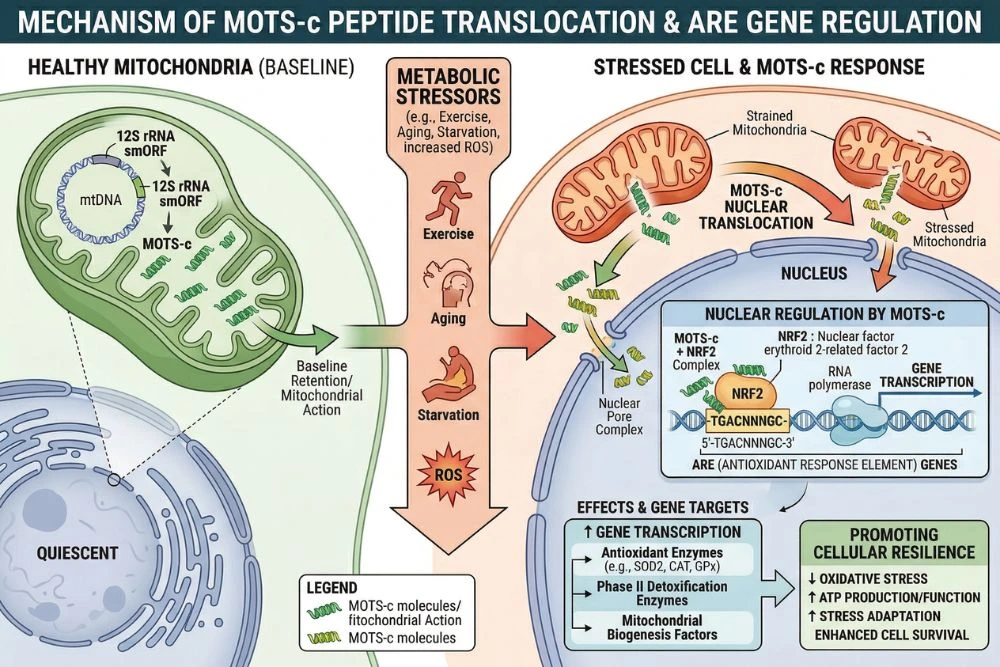
One of the most surprising aspects of MOTS-c biology is that it does not stay in the cytoplasm. Under metabolic stress, MOTS-c physically moves from the mitochondria into the cell's nucleus. This nuclear translocation is dependent on AMPK activation and allows MOTS-c to directly interact with transcription factors inside the nucleus.
Inside the nucleus, MOTS-c has been shown to interact with ATF1, NRF2, and NRF1, transcription factors that regulate genes involved in antioxidant defense, stress response, and mitochondrial biogenesis. Genes containing antioxidant response elements (ARE) are particularly responsive to MOTS-c's nuclear activity.
This makes MOTS-c what researchers call a "retrograde signal." Most cellular communication runs from the nucleus outward, with nuclear genes directing mitochondrial activity. MOTS-c represents the reverse: a mitochondrial signal communicating back to the nucleus and reshaping gene expression based on what is happening in the cell's energy-producing machinery.
This feedback loop has significant implications for understanding how cells adapt to exercise, caloric restriction, fasting, and metabolic stress. Conditions that push mitochondria harder appear to upregulate MOTS-c expression and secretion, which then modifies gene expression to make cells more resilient and metabolically efficient.
If you are interested in the broader landscape of how peptides regulate metabolism at the systemic level, the Ultimate Guide to Peptides for Weight Loss covers the wider ecosystem of metabolic peptides and how they interact with fat storage, energy use, and body composition research.
How MOTS-c Compares to Other Metabolic Peptides
MOTS-c sits within a broader family of mitochondrial-derived peptides that includes humanin and six small humanin-like peptides (SHLPs). Each has distinct biological actions, but they share the common feature of being encoded in mtDNA and acting as systemic signaling molecules. Within this family, MOTS-c stands out for its specific role in skeletal muscle metabolism and its strong exercise-response profile.
Compared to other peptides studied for metabolic effects:
- vs. Humanin: Humanin is primarily known for neuroprotective and anti-apoptotic effects. MOTS-c is more narrowly focused on metabolic regulation in muscle and adipose tissue, though both show exercise-induced increases.
- vs. Tesamorelin: Tesamorelin is a growth hormone-releasing hormone analog with FDA approval for visceral fat reduction. It works through the growth hormone/IGF-1 axis. MOTS-c works through the AMPK pathway and does not involve growth hormone signaling.
- vs. GLP-1 agonists: GLP-1 agonists like semaglutide act through the incretin system to improve insulin secretion and reduce appetite. MOTS-c acts directly on muscle metabolism independent of the incretin system.
For a deeper comparison of how research peptides differ from pharmaceutical metabolic compounds, the GLP-1 Agonists vs. Peptides guide provides useful context on the regulatory and mechanistic distinctions between the two categories.
MOTS-c is also connected to longevity research in ways that set it apart from most metabolic peptides. Studies have found elevated MOTS-c levels in centenarians compared to normally aging adults, suggesting a possible role in exceptional longevity. Whether MOTS-c directly contributes to longer life or simply reflects better mitochondrial health is an open and actively investigated question.
MOTS-c and the Broader Picture of Metabolic Aging
A comprehensive 2022 review published in the International Journal of Molecular Sciences (MDPI) examined MOTS-c's role across a range of aging-related conditions, including type 2 diabetes, cardiovascular disease, osteoporosis, postmenopausal obesity, and Alzheimer's disease. The review highlighted that the common thread across all these conditions is a gradual breakdown in mitochondrial metabolic balance, which is precisely where MOTS-c is active.
Some key findings from the aging and disease literature:
- Cardiovascular health: MOTS-c has been shown to reduce vascular calcification in animal models, reversing calcification-related reductions in AMPK phosphorylation.
- Bone health: MOTS-c promotes osteoblast proliferation and stimulates collagen synthesis through the TGF-beta/SMAD pathway, suggesting relevance to bone density maintenance.
- Adipose tissue: MOTS-c promotes conversion of white adipose tissue to brown adipose tissue and activates brown fat thermogenesis, which increases calorie expenditure at rest.
- Inflammatory response: By activating SIRT1 through the AMPK pathway, MOTS-c reduces expression of pro-inflammatory cytokines, a mechanism relevant to both metabolic disease and aging-related chronic inflammation.
This multi-system profile makes MOTS-c interesting not as a single-target compound but as a broad metabolic signal that appears to degrade in parallel with many of the processes associated with unhealthy aging. Research into whether restoring MOTS-c levels can functionally reverse any of these declines is ongoing, with animal data consistently positive and human data still accumulating.
For researchers who want broader context on how peptides fit into the longevity and cellular health space, the Future of Peptide Science: Trends in Regenerative Medicine provides a useful overview of where the field is heading.
Research Administration: What Scientists Need to Know
MOTS-c is not approved for human therapeutic use by the FDA or any major regulatory body. It is classified as a research compound, and all work with MOTS-c is conducted in preclinical research settings. Understanding the research context clearly is important for anyone working in this space.
Form and Reconstitution
Research-grade MOTS-c is typically supplied as a lyophilized (freeze-dried) powder. Standard reconstitution uses sterile bacteriostatic water or sterile saline, with acetic acid sometimes added at very low concentrations (0.1%) to aid dissolution. Most researchers prepare MOTS-c at concentrations between 1 and 5 mg/mL for animal studies, though exact concentrations vary by research protocol.
Stability Considerations
Lyophilized MOTS-c is relatively stable when stored at -20 degrees Celsius and kept away from repeated freeze-thaw cycles. Once reconstituted, solutions are typically used within 30 days when refrigerated and handled with appropriate sterile technique. As with all peptide research compounds, proper cold chain management from vendor to lab is critical for maintaining activity.
Sourcing for Research
When sourcing MOTS-c for research purposes, purity and verification are essential. Research-grade MOTS-c should come with a Certificate of Analysis (CoA) confirming identity, purity (typically above 98% for serious research), and absence of endotoxins and contaminants. If you are evaluating a source, the guide to How to Read a Certificate of Analysis provides a clear breakdown of what to look for.
Reputable vendors supplying research-grade MOTS-c can be found through PeptidesFinder. We may earn a commission from purchases made through links on this site, and we only feature vendors whose quality standards and documentation practices we consider credible for legitimate research use.
For accurate dosing calculations in research protocols, a peptide calculator is an essential tool for converting between mg/kg dosing, solution concentrations, and injection volumes.
Frequently Asked Questions
What does MOTS-c stand for?
MOTS-c stands for Mitochondrial Open Reading Frame of the 12S rRNA-c. The name describes exactly where in the mitochondrial genome the peptide is encoded: within the 12S ribosomal RNA region. It was named this way because when researchers discovered the short open reading frame hidden inside this region, they labeled it "c" to distinguish it from other potential reading frames in the same area.
Is MOTS-c a natural peptide in the human body?
Yes. MOTS-c is endogenous, meaning the human body produces it naturally. It is encoded in mitochondrial DNA and circulates in human blood at measurable concentrations. Levels vary by age, metabolic health, and physical activity status. The fact that it is naturally present in the body is one reason researchers are interested in it: studying how MOTS-c levels change under different conditions helps clarify its biological role, and understanding that role opens potential avenues for research into metabolic and age-related diseases.
How does MOTS-c relate to exercise?
Exercise is one of the most reliable triggers for MOTS-c production. Physical training, particularly endurance exercise, increases plasma MOTS-c levels. Conversely, administering MOTS-c to sedentary animals produces many of the same metabolic changes that exercise training produces: improved insulin sensitivity, enhanced glucose uptake in muscle, increased mitochondrial biogenesis, and better physical performance. This overlap is why MOTS-c has been described as an "exercise mimetic." It does not fully replicate everything exercise does, but it appears to activate many of the same downstream signaling pathways.
What is the AMPK pathway and why does MOTS-c activate it?
AMPK stands for AMP-activated protein kinase. It is a sensor molecule inside cells that detects low energy states and responds by switching the cell into an energy-conserving, fuel-burning mode. When MOTS-c inhibits the folate cycle, it causes AICAR to accumulate. AICAR directly activates AMPK, which then promotes glucose uptake, fatty acid oxidation, and mitochondrial biogenesis. This pathway is the same one activated by fasting, exercise, and metformin, making MOTS-c a naturally encoded activator of one of the body's most important metabolic regulatory systems.
Do MOTS-c levels decline with age?
Yes. Research consistently shows that circulating MOTS-c decreases as people age, and this decline tracks with the onset of insulin resistance, muscle loss, and metabolic dysfunction associated with normal aging. The decline appears to correlate with falling mitochondrial function more broadly, since MOTS-c production depends on healthy, active mitochondria. This age-related decline is one of the reasons researchers are investigating whether restoring MOTS-c levels could functionally slow aspects of metabolic aging. Animal studies using late-life MOTS-c administration have shown improvements in physical capacity and healthspan markers even when treatment begins at advanced ages.
Is MOTS-c approved for human use?
No. MOTS-c is not approved for human therapeutic use by the FDA or any other major regulatory agency. It is a research compound studied exclusively in preclinical settings. All published human data on MOTS-c consists of observational studies measuring naturally occurring levels rather than intervention trials. Any use of MOTS-c outside of properly authorized research protocols would be outside the scope of current scientific and regulatory standards. Researchers working with MOTS-c should do so under appropriate institutional oversight and ethical review.
How is MOTS-c different from other mitochondrial peptides like humanin?
MOTS-c and humanin are both encoded in mitochondrial DNA, but they have different primary actions. Humanin is known mainly for neuroprotection and anti-apoptotic effects, with the strongest research focus on its role in Alzheimer's disease models and cellular survival under stress. MOTS-c is more specifically tied to metabolic regulation in skeletal muscle and adipose tissue, with strong connections to insulin sensitivity, AMPK activation, and exercise adaptation. The two peptides can work together, and both levels are responsive to exercise training, but they act on different receptors and pathways.
Conclusion
MOTS-c is one of the most compelling discoveries to emerge from mitochondrial biology in the last decade. It is not just another metabolic peptide. It is proof that mitochondria, long described simply as the cell's powerhouse, are active participants in signaling between tissues, coordinating energy use, and regulating the biological machinery that determines metabolic health and physical capacity.
The research base is still growing. What is already clear is that MOTS-c rises with exercise, declines with age, activates AMPK through an elegant biochemical cascade, improves insulin sensitivity and physical performance in animal models, and appears to be part of the biological story behind why exercise is so protective against metabolic disease and aging. Whether the human data will eventually match the animal data at a therapeutic level is still an open question, but the early signals are strong.
For researchers in metabolic biology, exercise physiology, or aging science, MOTS-c represents a genuinely novel angle worth following. The mitochondrial genome was supposed to be fully understood. MOTS-c was the proof that it was not, and the field has been working through the implications of that ever since.
Interested in exploring more peptides shaping the future of metabolic research? The Peptides for Fat Loss guide covers the broader landscape of research compounds studied for their effects on body composition and metabolic function.
To stay at the forefront of emerging research in this space, follow FOXO4-DRI: The Senolytic Peptide for Reversing Aging and 5-Amino-1MQ: How to Block the Enzyme That Stores Fat, two upcoming research guides covering other exciting areas of metabolic and aging science.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. MOTS-c is sold as a research chemical and is not FDA-approved for human use. Research peptides are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any research compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.