FOXO4-DRI: The Senolytic Peptide for Reversing Aging
Discover how FOXO4-DRI targets senescent zombie cells by disrupting FOXO4-p53, restoring tissue health and reversing age-related cellular decline in research.
Most approaches to aging try to slow it down. A new class of compounds is taking a more direct approach: finding the cells responsible for age-related tissue damage and eliminating them entirely. FOXO4-DRI is one of the most precisely engineered compounds in this space. Rather than broadly suppressing inflammation or boosting antioxidants, it targets a specific protein interaction that keeps damaged cells alive when they should be dead. The result, in animal research, has been striking improvements in physical function, tissue health, and markers of biological aging.
This guide covers what FOXO4-DRI is, why senescent cells matter so much to aging research, exactly how FOXO4-DRI works at the molecular level, what the published research has found, and what scientists studying this compound need to understand about its current status and limitations.
The Problem: Senescent Cells and Why They Drive Aging
To understand FOXO4-DRI, you first have to understand senescent cells, because this peptide was designed to solve a very specific problem they create.
Every cell in your body has a built-in self-destruct mechanism called apoptosis. When a cell suffers serious DNA damage, gets too old, or stops functioning properly, this mechanism is supposed to activate and eliminate the cell cleanly. The cellular debris gets recycled, and the surrounding tissue replaces the lost cell with a healthy one. This is normal biology, and it works well when the system is running correctly.
But some damaged cells escape apoptosis. Instead of dying, they enter a state called cellular senescence. They stop dividing permanently and lose most of their normal function. They are not quite alive in the way a healthy cell is, and they are not quite dead either. Researchers sometimes call them zombie cells, because they just linger in tissue without contributing anything useful.
If that was all senescent cells did, they might not be such a problem. But they are metabolically active, and they continuously secrete a toxic mixture of inflammatory molecules. This secretory profile is called the SASP, which stands for senescence-associated secretory phenotype. A comprehensive review published in PMC on cellular senescence and therapeutic opportunities describes how the SASP contributes to the chronic sterile inflammation that underlies most major age-related diseases, including cardiovascular disease, metabolic dysfunction, cancer progression, and neurodegeneration.
A small number of senescent cells is normal and even beneficial early in life, where they play roles in wound healing and tumor suppression. The problem is that their numbers increase with age, immune clearance of these cells becomes less efficient over time, and the cumulative SASP burden eventually begins to actively degrade healthy surrounding tissue. When senescent cells reach a certain threshold in a given tissue, the local environment changes enough to drive dysfunction, organ aging, and the diseases associated with getting old.
The foundational research on SASP characterization, published in PMC and widely cited as the definitive SASP reference, established that senescent fibroblasts secrete dozens of inflammatory cytokines, chemokines, and matrix-remodeling proteases that alter the tissue environment around them, promote tumor growth in neighboring cells, and accelerate the aging of nearby healthy tissue.
This background makes the research question behind FOXO4-DRI obvious: if you could selectively remove senescent cells while leaving healthy cells untouched, could you reverse some of the damage they have been accumulating? The Baar et al. team at Erasmus University Medical Center Rotterdam decided to find out.
What Is FOXO4-DRI? Engineering a Precise Senolytic
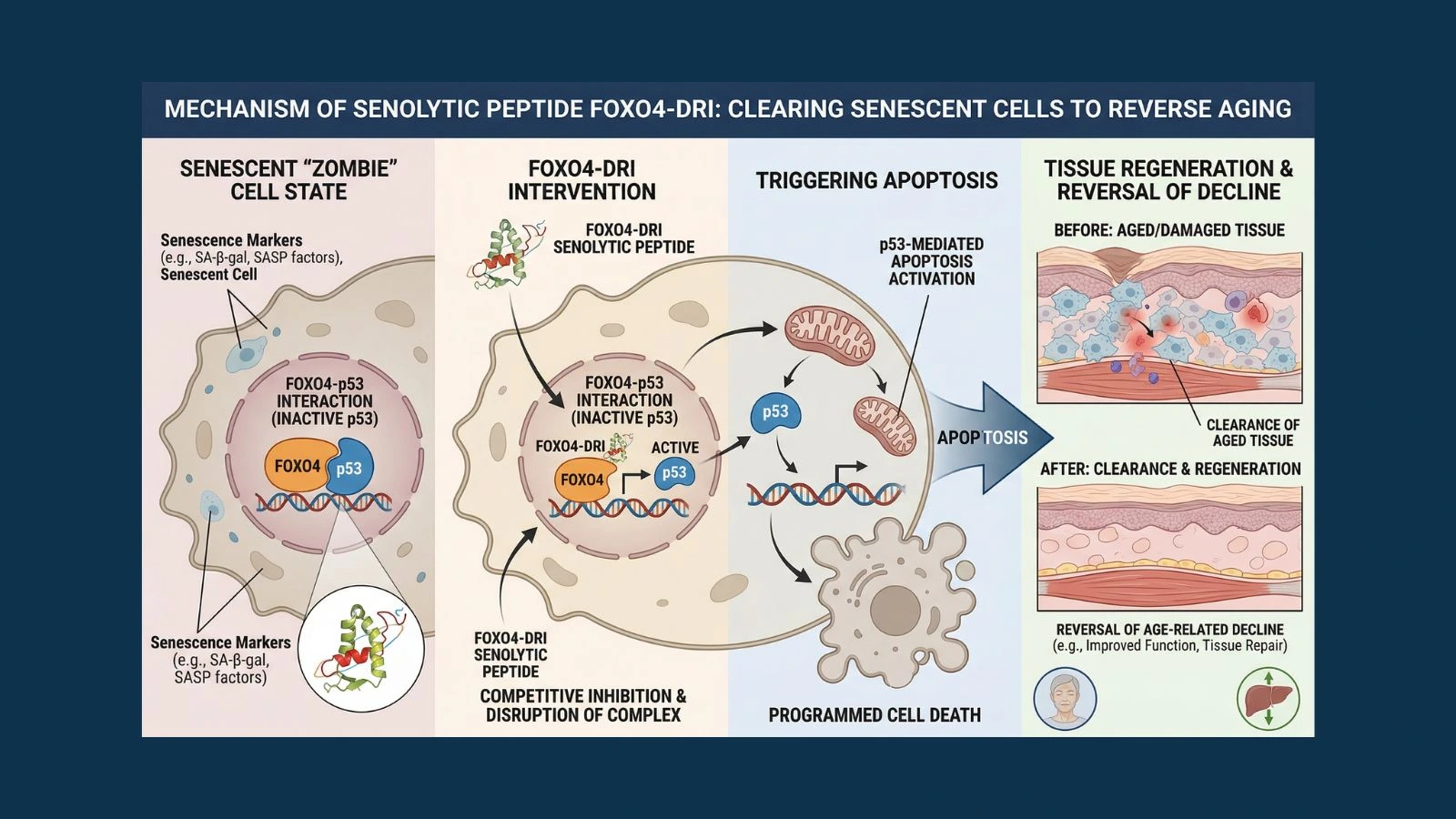
FOXO4-DRI stands for FOXO4 D-Retro-Inverso. The name contains the key to what makes this compound work.
FOXO4 is a transcription factor, a protein that controls which genes get turned on and off in a cell. In senescent cells, FOXO4 becomes concentrated in a specific nuclear compartment called the promyelocytic leukemia body. Inside these structures, FOXO4 physically binds to p53, one of the most important tumor suppressor and apoptosis-regulating proteins in biology. By grabbing onto p53 and keeping it trapped in the nucleus, FOXO4 prevents p53 from doing something it would otherwise do: trigger the apoptosis cascade that would kill the damaged cell.
In other words, the FOXO4-p53 interaction is the molecular handcuff keeping zombie cells alive. Break that interaction and p53 is released to do its normal job: travel to the mitochondria and initiate apoptosis.
The research team at Erasmus designed a peptide that mimics the p53-binding region of FOXO4. This peptide competes with endogenous FOXO4 for p53 binding. When the peptide wins that competition, p53 is freed from the nucleus, travels to the mitochondria, and triggers caspase-mediated apoptosis in the senescent cell.
The D-Retro-Inverso modification is what makes the compound viable as a research tool. Natural peptides are made from L-amino acids and are quickly degraded by enzymes in biological systems. By constructing the peptide from D-amino acids in a reversed sequence (retro-inverso), the researchers created a compound with the same three-dimensional binding shape as the original sequence but dramatically increased resistance to proteolytic breakdown. This D-form also improves cell penetration, which is critical since the peptide needs to reach FOXO4 inside the nucleus.
The landmark 2017 paper, published in Cell by Baar and colleagues and available through PubMed (PMID 28340339), established three crucial properties of FOXO4-DRI: it kills senescent cells, it spares normal proliferating cells, and it works in living animals under well-tolerated conditions. This combination of selectivity and in vivo efficacy is what set it apart from earlier approaches to senescent cell clearance.
How FOXO4-DRI Works: The Molecular Mechanism Step by Step
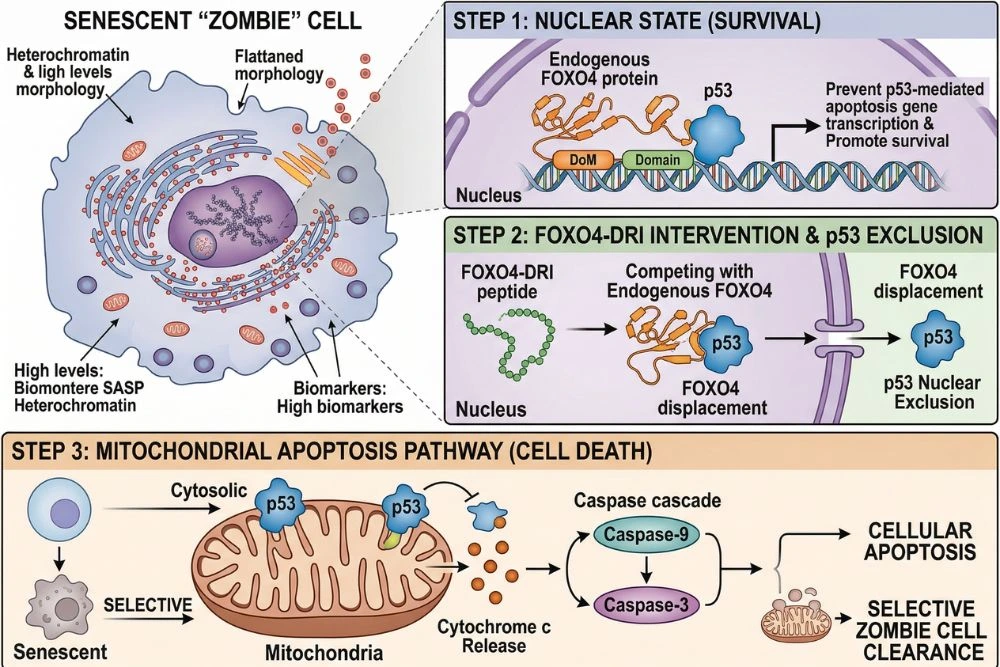
The mechanism of FOXO4-DRI is unusually elegant for a therapeutic compound. Most drugs work by blocking an enzyme or receptor. FOXO4-DRI works by mimicking one side of a protein-protein interaction to disrupt a lock-and-key relationship that only exists in senescent cells. Here is the full sequence of events:
Step 1 - Cell entry: FOXO4-DRI is a cell-penetrating peptide. Its D-retro-inverso structure gives it the ability to cross cell membranes and enter the cytoplasm without requiring a receptor or transport protein.
Step 2 - Competition for p53: Inside the cell, FOXO4-DRI travels to the nucleus where it competes with endogenous FOXO4 for binding to p53. In senescent cells, FOXO4 is highly expressed, and the FOXO4-p53 interaction is a major survival signal for these cells.
Step 3 - p53 nuclear exclusion: When FOXO4-DRI successfully outcompetes FOXO4 for p53 binding, p53 is released from the promyelocytic leukemia bodies where it has been sequestered. Without FOXO4 holding it in place, p53 undergoes nuclear exclusion, meaning it is expelled from the nucleus.
Step 4 - Mitochondrial translocation: Excluded p53 does not remain in the cytoplasm. It travels to the mitochondria, where it initiates a transcription-independent apoptosis cascade. This pathway activates the caspase enzymes that execute programmed cell death.
Step 5 - Selective apoptosis: In normal, non-senescent cells, FOXO4 is not significantly expressed and the FOXO4-p53 interaction does not exist at high levels. This means FOXO4-DRI has nothing meaningful to disrupt in healthy cells, which is why the compound shows selectivity for senescent cells over normal tissue.
This selectivity is the central advantage of FOXO4-DRI over earlier senolytics. Compounds like navitoclax (ABT-263) clear senescent cells by inhibiting BCL-2 family proteins, which are broadly expressed. This approach works but causes significant side effects including thrombocytopenia (dangerous reduction in platelets). FOXO4-DRI's target, the FOXO4-p53 interaction, is much more specific to senescent cells, which is why the original study reported that it was well tolerated in animals even with prolonged administration.
The 2017 Discovery: What the Research Found
The Baar et al. 2017 study in Cell remains the definitive evidence base for FOXO4-DRI. The research team tested the compound across multiple contexts: in vitro with human and mouse senescent cells, in a chemotherapy-induced accelerated aging model, and in naturally aged mice.
Selective Killing of Senescent Cells In Vitro
The researchers first established that FOXO4 is enriched in senescent cells compared to normal proliferating cells. Using human IMR90 fibroblasts induced to senesce by ionizing radiation, they showed that FOXO4 localizes to DNA-SCARS (DNA segments with chromatin alterations reinforcing senescence), where it forms a complex with p53.
When FOXO4-DRI was applied to cultures containing both senescent and normal cells, it selectively killed senescent cells while leaving normal proliferating cells unaffected. Cell viability measurements confirmed that the peptide induced p53-dependent apoptosis specifically in cells expressing the FOXO4-p53 interaction pattern characteristic of senescence.
Reversing Chemotherapy-Induced Aging
One of the most immediately clinically relevant findings was FOXO4-DRI's ability to counteract chemotoxicity. Doxorubicin is a commonly used chemotherapy drug with well-known long-term side effects including accelerated aging of multiple tissues, largely because chemotherapy induces widespread cellular senescence as a side effect of killing cancer cells.
Mice treated with doxorubicin and then given FOXO4-DRI showed significant recovery. Body weight returned toward normal. Liver damage markers (aspartate aminotransferase) decreased. Physical fitness, measured by grip strength and mobility testing, improved compared to doxorubicin-treated controls that did not receive the peptide. This suggests a potential application in oncology for managing the long-term toxic burden of chemotherapy.
Reversing Natural Aging in Old Mice
The most striking results came from naturally aged mice. Older mice receiving FOXO4-DRI showed improvements across several hallmarks of biological aging:
- Physical fitness measured by treadmill running and grip strength increased significantly compared to untreated aged controls
- Fur density and coat quality, which declines with age in mice as hair follicle stem cell function degrades, improved substantially
- Kidney function, measured by serum creatinine and urea nitrogen, improved in both fast-aging and naturally aged mice
- Renal tubular senescence, confirmed by SA-beta-galactosidase staining (the standard senescence marker), was reduced
The treatment was administered as an intraperitoneal injection three times per week for up to ten months. No obvious adverse effects were observed across this extended treatment period, which is a meaningful safety signal even in a mouse model.
The full text of the PMC article is available at PMC5556182, and contains the complete methodology, raw data, and supplementary analysis for researchers who want to examine the protocols directly.
FOXO4-DRI Beyond the Discovery Study: Expanding Applications
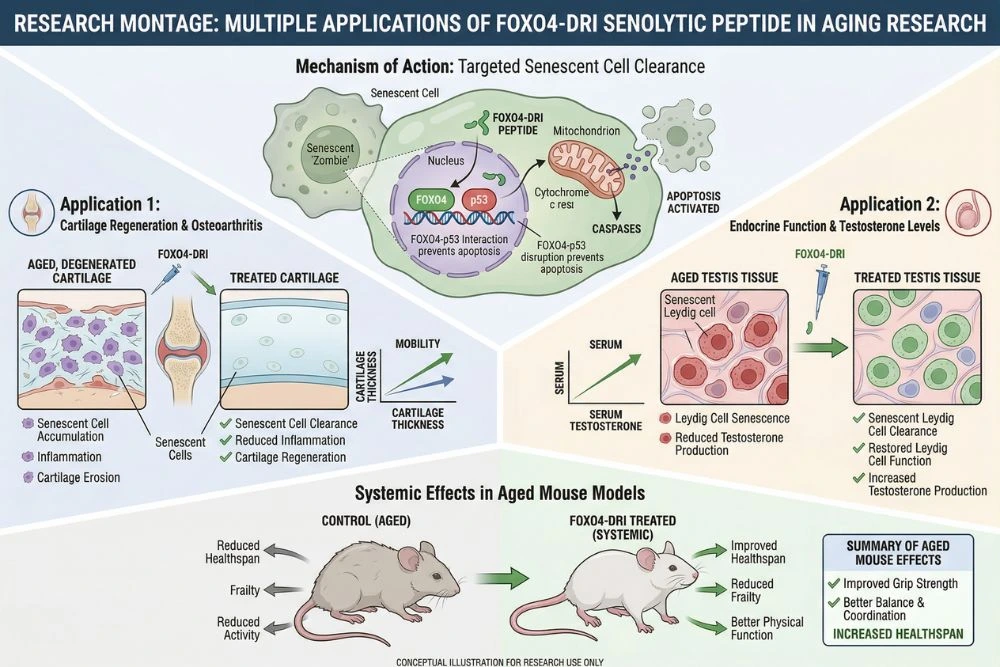
The Baar 2017 paper opened a significant research program. In the years since, multiple independent teams have tested FOXO4-DRI in different tissue contexts and aging-related conditions.
Testosterone Restoration and Leydig Cell Senescence
One particularly compelling application involves male hormonal aging. Testosterone production in men declines with age, partly because Leydig cells in the testes become senescent and lose their steroidogenic function. Standard hormone replacement therapy works around the problem by supplying exogenous testosterone, but it does not address the underlying cellular cause.
A 2020 study published in Aging (Albany) and available through PMC7053614, investigated whether clearing senescent Leydig cells with FOXO4-DRI could restore testosterone production naturally. The researchers found that FOXO4 expression in human testes increases with age and is associated with decreased testosterone synthesis. In aged mice, FOXO4-DRI selectively induced apoptosis in senescent Leydig cells while sparing the normal Leydig cell population. Following treatment, testosterone levels in the blood plasma of aged mice rose significantly compared to untreated controls. The testicular microenvironment, measured by inflammatory markers and cell density, also improved. This study expanded the potential relevance of FOXO4-DRI from general aging to specific age-related hormonal dysfunction.
Cartilage Regeneration and Joint Health
Another research direction involves cartilage repair. Autologous chondrocyte implantation (ACI) is a procedure where a patient's cartilage cells are harvested, expanded in culture outside the body, and then reimplanted into a damaged joint. The problem is that during in vitro expansion, chondrocytes accumulate senescent cells, and these senescent cells impair the quality of the cartilage formed after reimplantation.
A 2021 study published in Frontiers in Cell and Developmental Biology, with the full text available via PMC8116695, tested FOXO4-DRI as a pre-treatment for expanded chondrocytes before implantation. The compound removed more than half of the senescent cells in the expanded population without affecting the viability of non-senescent chondrocytes. The treated cartilage cultures showed significantly lower expression of SASP-related inflammatory secretory factors. While chondrogenic differentiation outcomes were mixed and need further investigation, the study confirmed FOXO4-DRI's senolytic selectivity in human cells and pointed toward potential applications in regenerative medicine.
The Broader Senolytic Landscape
A 2025 review published in PMC on targeting cellular senescence for healthy aging, places FOXO4-DRI within the broader field of senolytics, which includes compounds like dasatinib plus quercetin (the most clinically advanced combination currently in human trials), navitoclax, and the newer generation of rationally designed peptide senolytics. The review highlights that FOXO4-DRI's protein-protein interaction mechanism gives it a selectivity advantage over small-molecule senolytics that target broadly expressed survival proteins, and it is categorized as one of the benchmark agents against which new senolytic peptide candidates are measured.
How FOXO4-DRI Fits Into the Longevity Peptide Landscape
FOXO4-DRI occupies a distinct niche within the broader space of peptides studied for aging-related research. Most longevity-focused peptides work by optimizing existing biological processes: boosting growth hormone signaling, improving mitochondrial function, activating stress-resistance pathways. FOXO4-DRI takes a different approach entirely. Rather than enhancing cellular function, it removes cells that have become a liability to the system.
This is the difference between maintaining a machine and removing broken parts. Other peptides tune the engine. FOXO4-DRI removes the rust.
Compared to other well-studied research peptides in the aging space:
- vs. Epithalon: Epithalon works through telomerase activation, extending the replicative lifespan of cells. FOXO4-DRI takes the opposite approach by removing cells that have already reached or exceeded their replicative limit.
- vs. MOTS-c: MOTS-c improves mitochondrial metabolism and AMPK signaling, making existing cells work better. FOXO4-DRI targets the consequences of accumulated cellular dysfunction rather than metabolic efficiency.
- vs. Thymosin Alpha 1: Thymosin Alpha 1 boosts immune function to help the body do more of what it already does. FOXO4-DRI specifically addresses a failure mode of the immune system, the inadequate clearance of senescent cells that immune function can no longer handle efficiently in older organisms.
These compounds are not necessarily alternatives to each other. Researchers studying aging biology often consider multi-pathway approaches because aging involves simultaneous failure across multiple systems. For context on how different research peptides approach longevity-related biology from complementary angles, the Future of Peptide Science guide offers a useful overview of where the field is currently heading.
Research Considerations: What Scientists Need to Know
FOXO4-DRI is a research compound with no current FDA approval or equivalent for human therapeutic use. All published data comes from preclinical models. Understanding the research context clearly is essential for anyone working in this area.
Current Status and Limitations
While the animal data is compelling, several important limitations apply. First, all the major efficacy studies were conducted in mouse models. Mice age differently from humans, the distribution of FOXO4-expressing senescent cells may differ across tissues, and pharmacokinetic properties that work in mice do not always translate directly to larger organisms.
Second, senescence itself is not uniformly harmful. Senescent cells play constructive roles in wound healing and developmental processes. An ideal senolytic would discriminate not just between senescent and normal cells, but between beneficial and harmful senescent populations. FOXO4-DRI's selectivity is based on FOXO4 expression, which is enriched in senescent cells but may not perfectly map to all senescent cell subtypes.
Third, the long-term consequences of substantially reducing the total senescent cell burden in an organism over an extended period are not yet characterized in any model. The animal studies reported no obvious adverse effects, but comprehensive long-term safety data does not yet exist.
Form, Stability, and Reconstitution
Research-grade FOXO4-DRI is supplied as a lyophilized powder. Reconstitution typically uses sterile bacteriostatic water or PBS, with DMSO sometimes used in cell culture applications at appropriate low concentrations. The D-retro-inverso modification gives FOXO4-DRI significantly better stability than natural L-peptides, which is a practical advantage for storage and handling. Lyophilized material stored at -20 degrees Celsius maintains stability for extended periods, and reconstituted solutions should be aliquoted and stored at -80 degrees Celsius to prevent activity loss from freeze-thaw cycles.
Sourcing and Documentation
Research-grade FOXO4-DRI should be sourced from vendors who supply a full Certificate of Analysis confirming identity by mass spectrometry, purity above 95% (preferably 98%+), and endotoxin testing. If you are evaluating a source, the guide to How to Read a Certificate of Analysis walks through exactly what to look for when assessing peptide quality documentation.
Reputable research-grade FOXO4-DRI can be found through vendors listed on PeptidesFinder. We may earn a commission from purchases made through links on this site, and we only list vendors whose documentation and quality practices meet the standards required for legitimate preclinical research.
For accurate preparation of research solutions, a peptide calculator is essential for converting mg/kg doses to injection volumes based on your working stock concentration.
For researchers new to peptide-based research more broadly, the Beginner's Guide to Peptide Therapy provides a useful foundation before diving into more advanced compounds like FOXO4-DRI.
Frequently Asked Questions
What does FOXO4-DRI stand for?
FOXO4-DRI stands for FOXO4 D-Retro-Inverso. FOXO4 refers to the forkhead box O4 transcription factor, which is the protein this compound targets. D-Retro-Inverso describes the structural modification that makes the peptide functional as a drug: it is constructed from D-amino acids (mirror images of the natural L-amino acids found in the body) arranged in a reversed sequence. This modification gives the peptide the same three-dimensional shape needed to bind its target while making it resistant to the enzymes that would normally break down a natural peptide quickly.
What is a senolytic and why does it matter for aging?
A senolytic is any compound that selectively induces apoptosis in senescent cells while leaving healthy cells intact. Senescent cells accumulate with age, stop functioning properly, and continuously secrete inflammatory molecules through the SASP. This chronic low-level inflammation is strongly associated with most major age-related diseases including heart disease, diabetes, neurodegeneration, and cancer progression. Senolytics matter because they address one of the root biological causes of aging rather than just managing symptoms. By clearing the cells responsible for this toxic inflammatory environment, the goal is to restore tissue homeostasis to a state more like that seen in younger organisms.
How does FOXO4-DRI select senescent cells over healthy ones?
FOXO4-DRI's selectivity comes from a biological difference between senescent and normal cells. In senescent cells, FOXO4 accumulates in specific nuclear structures called promyelocytic leukemia bodies, where it physically binds p53 and prevents it from triggering apoptosis. This FOXO4-p53 interaction is what keeps zombie cells alive despite being damaged. Normal proliferating cells do not have this FOXO4-p53 interaction at significant levels, so FOXO4-DRI has no meaningful target in them. It disrupts what exists in senescent cells but not in healthy tissue, which is why the original study found that it kills senescent cells while leaving normal cells unaffected.
What were the key results in the 2017 Baar et al. discovery study?
The 2017 Baar et al. study published in Cell showed three major categories of results. First, FOXO4-DRI selectively killed senescent human and mouse fibroblasts in culture without harming normal proliferating cells, confirming its basic senolytic mechanism. Second, in mice treated with doxorubicin to induce accelerated aging from chemotherapy, FOXO4-DRI reversed many of the toxic effects: body weight recovered, liver damage markers fell, and physical fitness improved. Third, in naturally aged mice, treatment with FOXO4-DRI over extended periods improved physical fitness, restored fur density, and improved kidney function compared to untreated aged controls. No obvious adverse effects were observed across ten months of treatment.
Has FOXO4-DRI been tested in humans?
No human clinical trials of FOXO4-DRI have been published as of the current research record. All available efficacy data comes from in vitro cell culture studies and mouse models. The compound is categorized as a research chemical and is not approved by the FDA or any equivalent regulatory body for human therapeutic use. Human research on senolytic compounds is an active and growing field, but FOXO4-DRI specifically has not progressed to formal clinical trials. Researchers working with this compound do so exclusively in properly authorized preclinical research settings.
What is the SASP and why is it harmful?
SASP stands for senescence-associated secretory phenotype. It is the collection of inflammatory molecules that senescent cells continuously secrete into their surrounding tissue environment. The SASP includes pro-inflammatory cytokines such as IL-6 and IL-8, matrix metalloproteinases that degrade structural tissue, growth factors that can promote abnormal cell behavior in neighboring tissue, and chemokines that recruit immune cells. In small amounts or for short periods, the SASP has beneficial functions in wound healing and immune signaling. But as senescent cells accumulate with age and the SASP becomes chronic, it creates a persistent low-level inflammatory state throughout the body, which is a major driver of the tissue dysfunction and disease risk that characterizes biological aging.
How does FOXO4-DRI compare to other senolytics like dasatinib and quercetin?
Dasatinib and quercetin are the most clinically advanced senolytic combination currently in human research and represent a different mechanistic approach. Dasatinib targets multiple tyrosine kinases that senescent cells depend on for survival, while quercetin has broad anti-inflammatory and senomorphic properties. This combination is effective but affects multiple targets simultaneously, which creates more potential for off-target effects. FOXO4-DRI is more targeted: it disrupts a single specific protein-protein interaction (FOXO4-p53) that is particularly enriched in senescent cells. In preclinical studies, this specificity translates to a cleaner safety profile. The trade-off is that FOXO4-DRI is a peptide requiring injectable administration and is less bioavailable than small molecules like dasatinib. Both approaches are actively studied and may eventually prove complementary rather than competitive.
Conclusion
FOXO4-DRI represents one of the most targeted and mechanistically precise tools in aging biology research. It does not attempt to broadly slow down cellular aging or boost a non-specific protective pathway. Instead, it identifies a specific protein interaction that senescent cells depend on for survival, disrupts that interaction with high selectivity, and triggers the cells' own built-in death machinery to clear them from the tissue.
The results in animal models have been genuinely impressive: restored physical function, improved organ health, better hair growth, and reversal of hormonal decline across multiple independent research groups. Whether these effects will translate to human biology at a clinically meaningful scale remains the central open question, and it is a question the field is actively working to answer.
For researchers in aging biology, senescence, or regenerative medicine, FOXO4-DRI offers a unique experimental tool. It allows targeted depletion of senescent cells in a way that can isolate their contribution to age-related tissue dysfunction from all the other processes happening simultaneously in aging organisms. That precision is exactly what the field needs to move from correlation to causation in understanding how senescence drives aging.
Interested in exploring how other research peptides approach aging-related biology? The Epithalon guide covers the telomere-focused approach to cellular longevity, and the Thymosin Alpha 1 guide examines how immune optimization intersects with healthy aging research.
Also watch for the upcoming deep dive into 5-Amino-1MQ: How to Block the Enzyme That Stores Fat, which explores another cutting-edge metabolic research compound approaching energy regulation from a completely different angle.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. FOXO4-DRI is sold as a research chemical and is not FDA-approved for human use. Research peptides are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in controlled environments with proper ethical oversight. This article does not constitute medical advice. Always consult a licensed healthcare provider before using any research compound. Never disregard professional medical advice or delay seeking it because of information you read online. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.