TB-500 Peptide: The Ultimate Guide to Injury Recovery and Flexibility
Discover how the TB-500 peptide uses thymosin beta-4 biology to accelerate injury recovery, reduce inflammation, boost flexibility, and restore damaged tissue.
TB-500: Research Overview and Molecular Mechanisms
When tissue gets injured, the body launches a repair process that is elegant in theory but frustratingly slow in practice. Cells need to migrate to the damage site, new blood vessels need to form, inflammation has to be controlled, and structural proteins need to be rebuilt. Each step depends on the one before it, and any bottleneck in the chain delays the whole process.
TB-500 peptide works by targeting one of the earliest and most important steps in that chain. It mimics a peptide your own body uses to prime cells for migration and coordinate the tissue repair response from the moment injury occurs. Understanding how it works, what the research says, and why it has attracted so much attention in injury recovery research starts with understanding the molecule it is based on: thymosin beta-4.
What Is TB-500? The Relationship Between TB-500 and Thymosin Beta-4
TB-500 is a synthetic research peptide derived from thymosin beta-4, a naturally occurring 43-amino-acid protein found in virtually every cell of the human body. The two are often used interchangeably in research discussions, though they are technically distinct: thymosin beta-4 is the full endogenous protein, while TB-500 specifically refers to the synthesized fragment based on its most biologically active region.
Thymosin beta-4 was first isolated from thymic tissue in the 1960s as part of a larger investigation into thymic hormones and their role in immune function. Over the following decades, researchers discovered that its biological role was far broader than originally thought. Rather than acting primarily as an immune regulator, thymosin beta-4 turned out to be one of the body's most important wound-healing and tissue-repair coordinators, functioning in virtually every tissue type and responding directly to injury.
Thymosin beta-4 is present in high concentrations inside cells, particularly in platelets, macrophages, and the tissues with the highest repair demands: the heart, lungs, spleen, and skeletal muscle. When tissue is damaged, platelets arriving at the injury site release thymosin beta-4 directly into the wound environment. This release signals the start of the repair cascade, making it one of the earliest molecular events in the healing process.
TB-500 as a research compound was synthesized to study and exploit this biology in preclinical research settings. The active fragment it is based on contains the central actin-binding domain of thymosin beta-4, which is responsible for the majority of the protein's tissue repair activity.
The Science of How TB-500 Works: Actin, Cell Migration, and Repair
The Actin Connection
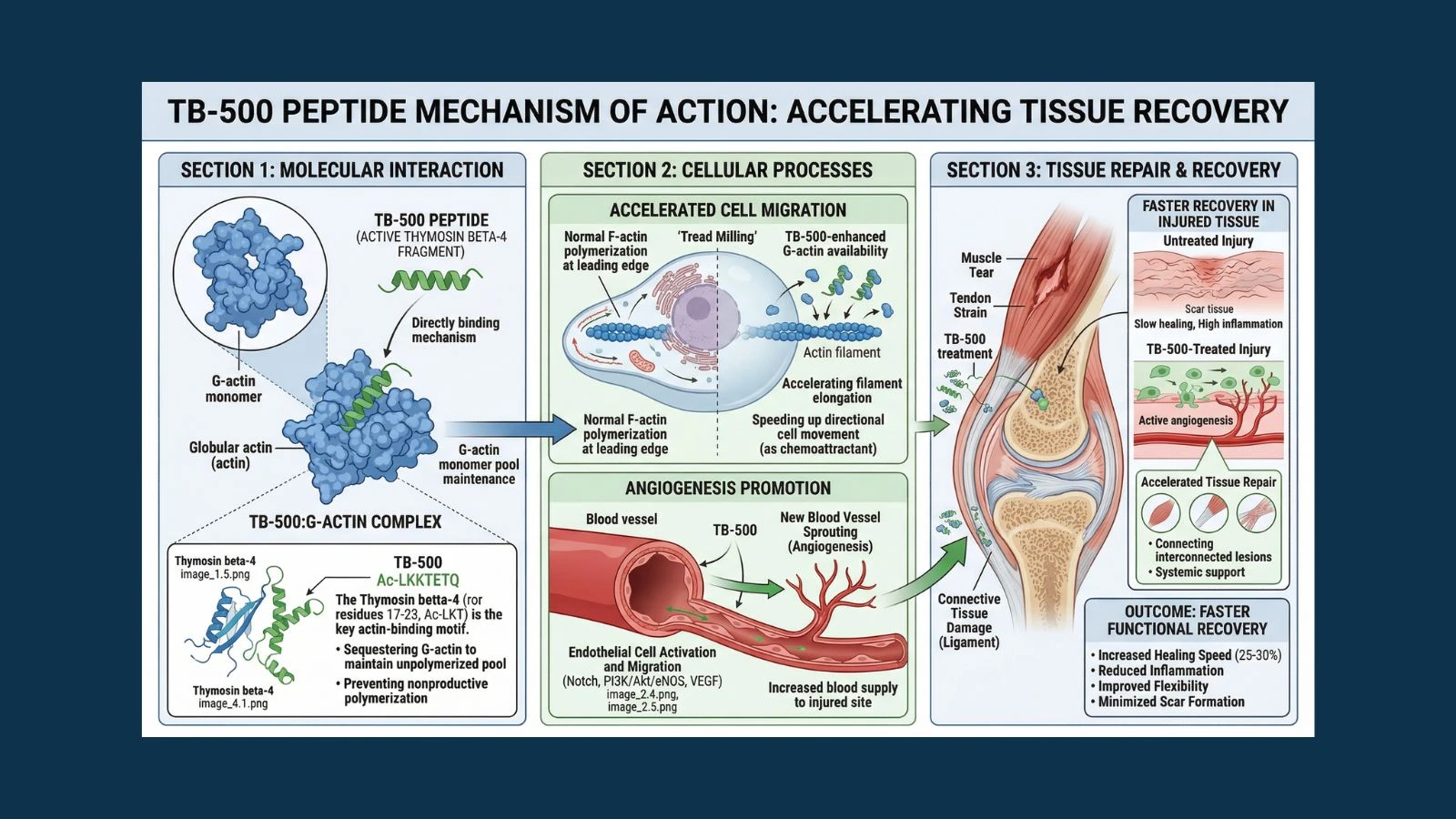
Actin is one of the most abundant proteins in the human body. Inside cells, it exists in two forms: G-actin (globular, monomeric) and F-actin (filamentous, polymerized). The ratio between these two forms determines whether a cell can move. When a cell needs to migrate, it polymerizes G-actin into F-actin filaments at its leading edge, creating the structural push that drives it forward.
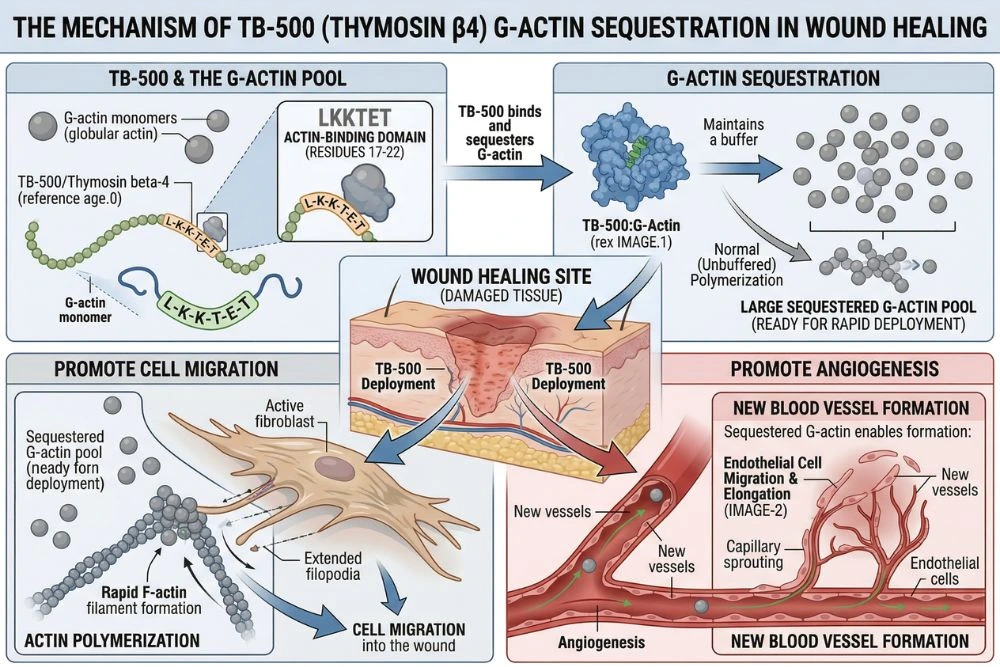
Thymosin beta-4 is the primary G-actin sequestering protein in mammalian cells. It holds a large reserve of G-actin monomers in a ready-to-polymerize state, essentially maintaining a loaded spring of actin that can be deployed rapidly when the cell receives a migration signal. Without this reserve, cells cannot respond quickly enough to injury signals to mount an effective repair response.
The landmark structure-function study published in PubMed Study 20179146 mapped the active sites on thymosin beta-4 responsible for its various biological effects. The central actin-binding domain, a seven-amino-acid sequence called LKKTETQ at residues 17 to 23, was identified as the key site driving angiogenesis, wound healing, and cell migration. This is the region that TB-500 is based on, explaining why a fragment of the full 43-amino-acid protein retains so much of its biological activity.
Angiogenesis: Building the Blood Supply for Repair
Effective tissue repair requires more than cell migration; it requires a blood supply. New blood vessels need to form to deliver oxygen, nutrients, and immune cells to the repair site while removing cellular waste. This process is called angiogenesis, and it is one of TB-500's most important documented activities.
A dedicated study of the angiogenic mechanism, published in PubMed Study 14500546, confirmed that the actin-binding motif of thymosin beta-4 is essential for its angiogenic activity. The researchers showed that thymosin beta-4 promotes endothelial cell migration, adhesion, tubule formation, and aortic ring sprouting. Even a seven-amino-acid fragment containing just the LKKTET motif retained near-identical angiogenic activity to the full protein, confirming that TB-500's truncated structure captures the functionally critical portion of the molecule.
Anti-Inflammatory Effects and the Ac-SDKP Domain
Beyond actin regulation and angiogenesis, thymosin beta-4 carries a second functional region at its amino terminus: a four-amino-acid sequence called Ac-SDKP. This site has documented anti-inflammatory activity. Research has shown that Ac-SDKP generally blocks inflammation and reduces fibrosis, while a slightly longer 15-amino-acid segment from the same terminus promotes cell survival and blocks apoptosis.
This dual functionality, promoting repair while simultaneously suppressing excessive inflammation, is part of what makes TB-500 biologically distinct from simpler growth factors that only drive proliferation. In injury settings, unchecked inflammation can delay healing and increase scar formation. TB-500's ability to moderate the inflammatory response while simultaneously driving the repair cascade addresses both sides of the problem.
A comprehensive review of thymosin beta-4's multi-functional role, published in PubMed Study 22074294, describes how these combined actions work together: after injury, thymosin beta-4 is released by platelets and macrophages to protect cells from further damage, reduce apoptosis and inflammation, promote cell migration and stem cell mobilization, stimulate angiogenesis, and reduce scar-forming myofibroblast activity.
TB-500 and Wound Healing: What the Research Shows
The most thoroughly studied application of thymosin beta-4 biology is wound healing, particularly dermal wound repair. Multiple animal models have consistently demonstrated accelerated healing, and the research has progressed far enough that phase 2 clinical trials have been conducted in human patients.
The foundational wound healing study, published in PubMed Study 10469335, documented increased reepithelialization, wound contraction, and collagen deposition in treated wounds compared to controls. These results were replicated and extended across other models. Research summarized in PubMed Study 23050815 showed acceleration of dermal healing in rats, mice, diabetic models, and aged animals. Clinical data in stasis and pressure ulcer patients showed healing roughly one month faster than controls, confirming relevance beyond preclinical research.
TB-500 for Muscle, Tendon, and Connective Tissue Repair
While wound healing research provides the most complete evidence base, TB-500's repair-promoting activities extend well beyond skin. The same mechanisms that drive dermal healing, cell migration, angiogenesis, and stem cell mobilization, are relevant to virtually every tissue type that suffers injury. Muscle, tendon, and connective tissue research has become one of the most active areas of TB-500 investigation.
Skeletal Muscle and Myoblast Recruitment
After skeletal muscle injury, satellite cells must migrate to initiate repair. Research shows thymosin beta-4 is upregulated in early skeletal muscle regeneration in mice and accelerates closure and chemotaxis of myoblasts. Myoblasts from adult mouse satellite cells migrate toward sulphoxidized thymosin beta-4, suggesting active recruitment by the endogenous peptide.
A recent review in PMC Therapeutic Peptides in Orthopaedics places TB-500 alongside BPC-157 and GHK-Cu as key peptides studied in orthopaedic contexts.
Tendon and Ligament Biology
Tendon and ligament injuries heal slowly because of limited blood supply and few resident cells. TB-500’s angiogenic activity improves vascularization, oxygenation, and nutrient delivery. Its Ac-SDKP-mediated anti-fibrotic effects help reduce disorganized scar tissue, improving tissue quality and functional recovery.
Flexibility and Range of Motion
TB-500 has also attracted research interest regarding flexibility and range of motion due to its effects on connective tissue remodeling. Its combination of anti-inflammatory, anti-fibrotic, and collagen-organizing activity suggests potential benefits in reducing stiffness and improving pliability, though direct flexibility data are limited.
TB-500 in Cardiac and Neurological Tissue Research
Cardiac Research
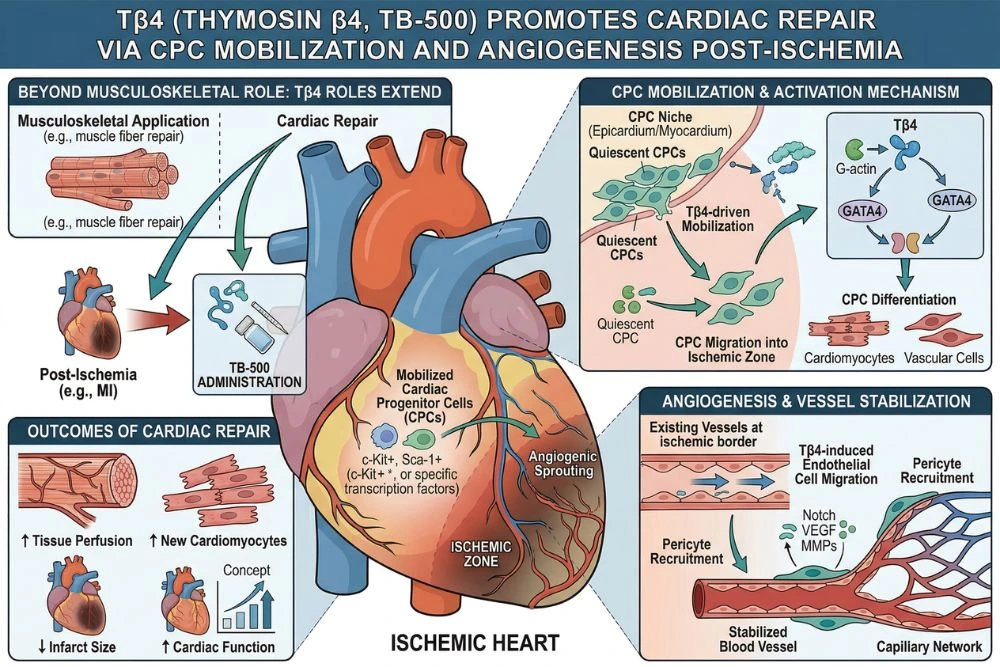
Heart muscle has limited regenerative ability post-injury. Thymosin beta-4 activates cardiac progenitor cells and promotes migration into damaged myocardial tissue. Studies show systemic administration increases progenitor cell count and improves function after induced cardiac ischemia. While preliminary, it represents an emerging area of regenerative biology.
Neurological and Corneal Research
Thymosin beta-4 has shown promise in spinal cord injury models, improving neuronal survival and reducing glial scarring. Corneal applications have been investigated by the FDA due to preclinical evidence of accelerated epithelial migration and wound closure, underscoring translational potential.
How TB-500 Compares to Other Recovery Peptides
TB-500 vs. BPC-157: Both promote tissue repair but differ mechanistically—BPC-157 acts via nitric oxide and integrin signaling, while TB-500 acts via actin dynamics and thymosin beta-4 biology. They are sometimes co-studied for complementary effects. See BPC-157 comprehensive research guide.
TB-500 vs. GHK-Cu: GHK-Cu focuses on collagen and enzyme regulation. TB-500 acts earlier, mobilizing cells and building blood supply. See GHK-Cu guide.
TB-500 vs. IGF-1 LR3: IGF-1 promotes growth factor signaling and satellite cell proliferation, while TB-500 promotes early repair through migration and angiogenesis—mechanistically sequential processes.
Research Considerations: What Scientists Working with TB-500 Need to Know
TB-500 is a research compound not approved for therapeutic use. Data come primarily from in vitro and animal studies, except for thymosin beta-4 wound healing trials using the full protein.
Form, Reconstitution, and Stability
Research-grade TB-500 is supplied as a lyophilized powder. Reconstitution uses sterile bacteriostatic water, typically 1–2 mg/mL. Stored at -20°C, it remains stable for 24 months; reconstituted solutions should be refrigerated and used within 30 days.
TB-500 in Veterinary Research
TB-500 has a documented history in equine medicine for tendon and ligament recovery. It is detectable by mass spectrometry in plasma and urine and is a prohibited substance in competitive equestrian sports.
Sourcing for Research
Always source TB-500 from vendors providing Certificates of Analysis confirming peptide identity, purity above 98%, and endotoxin testing. For guidance, see How to Read a Certificate of Analysis guide.
Use the peptide calculator to determine accurate research doses and solution volumes.
Frequently Asked Questions
What is TB-500 and how does it differ from thymosin beta-4?
TB-500 is a synthetic peptide based on the active fragment of thymosin beta-4, a naturally occurring 43-amino-acid protein found in nearly all cells. The key difference is that thymosin beta-4 is the full endogenous protein, while TB-500 is a standardized synthesized fragment containing its actin-binding domain. It retains most of thymosin beta-4's biological activity, enabling focused preclinical research.
How does TB-500 promote tissue repair?
TB-500 promotes tissue repair through three core mechanisms. It regulates actin to enable rapid cell migration, promotes angiogenesis to build blood supply, and suppresses excessive inflammation via its Ac-SDKP domain. Together these processes create ideal conditions for tissue recovery.
What tissue types has TB-500 been studied in?
TB-500 has been studied in dermal wounds, skeletal muscle, tendons, ligaments, cardiac tissue, neurological injury models, and corneal healing. Consistent results across these tissues reflect the universality of actin regulation and angiogenesis in biological repair.
Is TB-500 the same peptide used in equine racing?
Yes. TB-500 has been used in equine sports medicine and is listed as a prohibited substance in competitive racing due to its performance-enhancing potential. These veterinary studies contribute to understanding its pharmacokinetics and tolerability in large mammals.
What is the Ac-SDKP sequence and why does it matter?
Ac-SDKP is a four-amino-acid sequence with anti-inflammatory and anti-fibrotic properties. It inhibits cytokine production and inflammation, reducing poor-quality scar formation during tissue repair.
Can TB-500 be stacked with BPC-157 for research purposes?
TB-500 and BPC-157 are often co-studied because their mechanisms complement each other—TB-500 promotes migration and vascularization, while BPC-157 influences nitric oxide and extracellular matrix repair.
Has TB-500 been tested in human clinical trials?
Only thymosin beta-4, the parent molecule, has reached phase 2 clinical trials for wound healing applications, shortening healing time by about a month in chronic ulcers. TB-500 itself has not been tested in formal human trials.
Conclusion
TB-500 stands apart because its biological mechanisms—actin regulation, angiogenesis, inflammation control, and fibrosis reduction—are clearly defined and independently documented. Thymosin beta-4's phase 2 clinical data provide rare translational evidence in this research space. For scientists studying repair and recovery biology, TB-500 represents a well-characterized, versatile peptide with strong evidence and broad applicability.
Learn more about related peptides in the BPC-157 research guide and Ultimate Guide to Collagen Peptides.
Official Research Disclaimer
The information provided in this guide is for educational purposes only. TB-500 is sold as a research chemical and not approved for human use. Research peptides are intended for laboratory research only and not for human consumption or therapeutic application. Always consult a licensed professional before using any research compound. The author and publisher disclaim any liability arising from misuse or misinterpretation of this information.