Peptide Peptide Bonds: The Chemistry of Connection
What is a peptide bond? We explain the chemistry behind the peptide peptide connection, amide linkages, and how amino acids join to form life's building blocks.
Peptide Bonds: The Chemical Glue of Life
In the world of biochemistry, life is built on connections. From the enzymes that digest your food to the collagen that holds your skin together, every protein in your body relies on a specific type of chemical glue to stay intact.
This glue is called the peptide bond.
When researchers type "peptide peptide" into search engines, they are often looking for the fundamental mechanics of how these molecules attach to one another. How does one amino acid hold hands with the next? Why are these bonds so stable that they can survive boiling water, yet flexible enough to fold into complex enzymes?
This guide dives deep into the chemistry of connection. We will explore the amide linkage, the process of dehydration synthesis, and why the strength of these bonds determines the quality of the research peptides you buy.
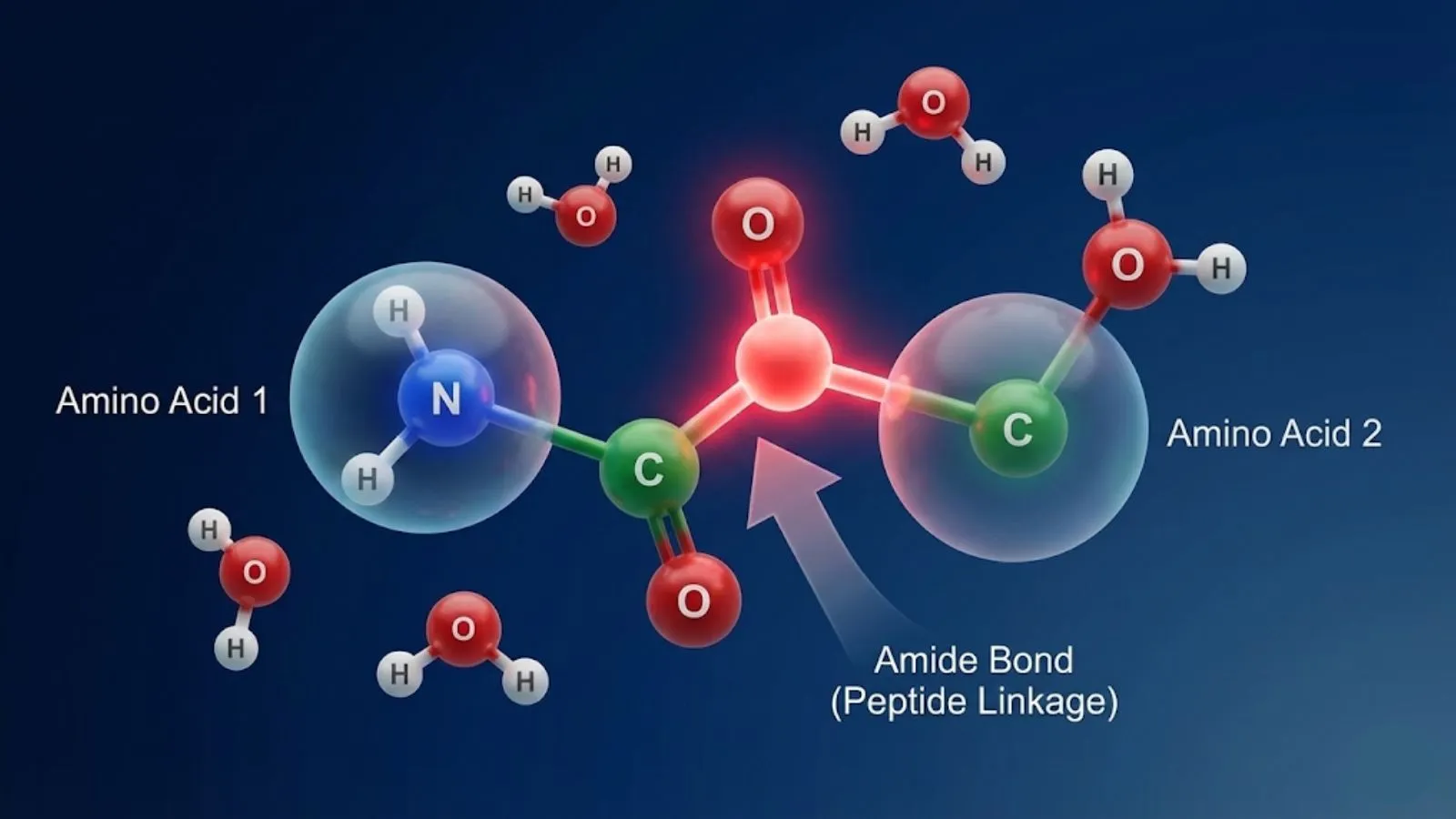
The Definition: What is a Peptide Bond?
A peptide bond is a specific type of covalent chemical bond. It links the carboxyl group (-COOH) of one amino acid to the amino group (-NH2) of another.
According to Britannica Science, this bond is chemically defined as an amide linkage. It is the "spine" of every protein molecule. Without it, life as we know it would be a puddle of loose amino acids.
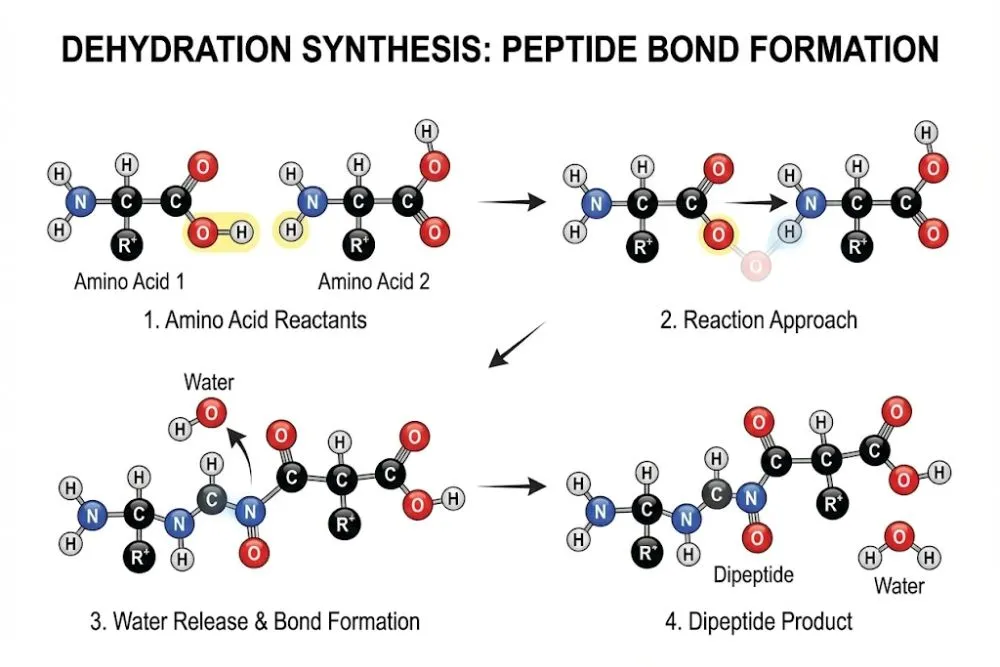
The Reaction: When two amino acids meet, they undergo a dehydration synthesis (or condensation) reaction. A water molecule (H2O) is removed, and the remaining carbon and nitrogen atoms snap together.
The Result: A dipeptide (two amino acids linked by one bond).
To understand how these chains grow from simple bonds into complex structures, read our guide on What Are Peptides? The Ultimate Beginner’s Guide.
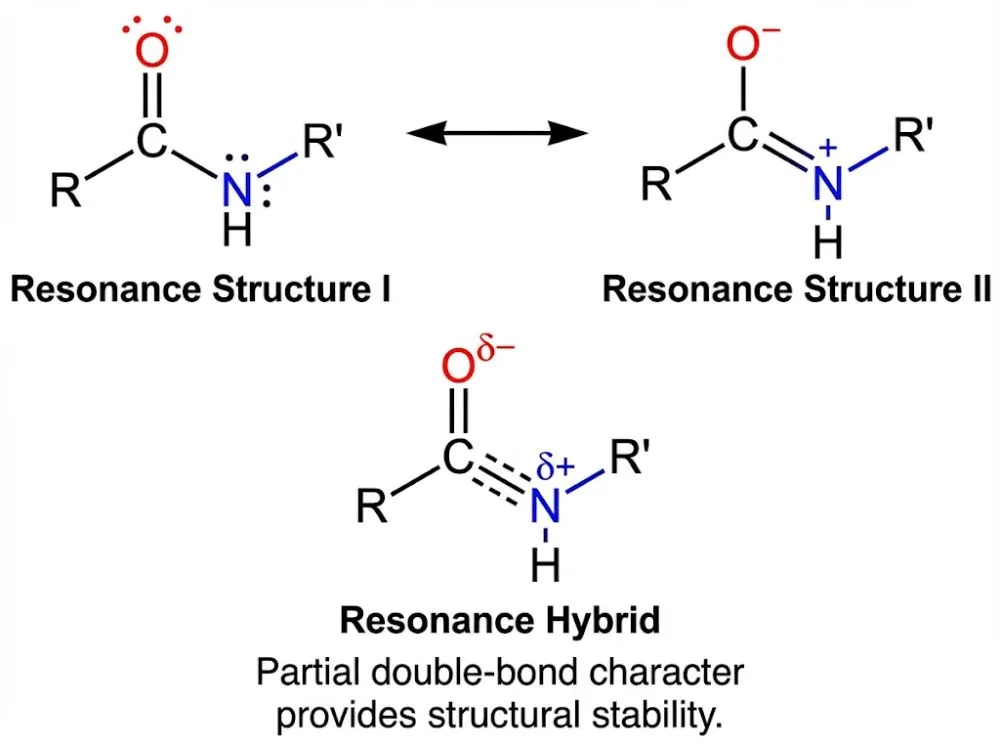
Why Is This Bond So Special? (The Resonance Factor)
The peptide bond isn't just a simple single bond; it has superpowers.
In a standard single bond, molecules can rotate freely, like a wheel on an axle. However, the peptide bond has partial double-bond character due to a phenomenon called resonance.
As explained in OpenStax Biology 2e, this resonance makes the bond rigid and planar (flat). This rigidity is crucial. If the bond were floppy, proteins would constantly lose their shape. Because it is stiff, it allows peptides to fold into stable, predictable structures, like the triple helix we discussed in Collagen Polypeptides.
Breaking the Bond: Hydrolysis
The bond is strong, but it isn't invincible. The process of breaking a peptide bond is called hydrolysis (literally "water breaking").
This is what happens in your stomach. Enzymes like pepsin add a water molecule back into the bond, snapping it open and releasing the individual amino acids so your body can absorb them.
In the lab, this stability is a double-edged sword.
Pros: It means lyophilized (freeze-dried) peptides are incredibly stable on the shelf.
Cons: Once reconstituted with water, the clock starts ticking. Over time, the water will naturally hydrolyze the bonds, degrading the peptide.
This is why proper storage is critical. Learn how to check if your peptides have degraded by reading Scientific Peptides: How to Read a Certificate of Analysis.
Polypeptide Formation: The Chain Reaction
The "peptide peptide" keyword often implies the connection of multiple units.
Dipeptide: 2 amino acids, 1 bond.
Tripeptide: 3 amino acids, 2 bonds.
Polypeptide: 50+ amino acids, 49+ bonds.
According to Molecular Biology of the Cell (NCBI Bookshelf), the process of Translation is what dictates this sequence. The ribosome (the cell's factory) reads the genetic information from mRNA and acts like a mechanical ratchet, stitching the peptide bonds together one by one to create the specific protein encoded by your genes.
The "Peptide-Peptide" Interaction in Drugs
In modern pharmacology, scientists are now designing drugs that mimic these bonds.
Many "peptidomimetics" (drugs that mimic peptides) replace the natural peptide bond with a chemical substitute that is harder for enzymes to break. This extends the half-life of the drug. For example, modifying these bonds is how researchers transformed fleeting natural hormones into long-lasting drugs like Semaglutide.
Frequently Asked Questions
What is the difference between a peptide bond and a covalent bond?
A peptide bond is a type of covalent bond. "Covalent" is the broad category (sharing electrons), while "Peptide Bond" is the specific name for the amide link connecting a carbon atom of one amino acid to the nitrogen atom of another.
Does boiling water break peptide bonds?
Generally, no. Peptide bonds are kinetically stable. Boiling them in water (denaturation) might unfold the protein (break the weak hydrogen bonds), but it usually won't snap the primary peptide bonds. You need a strong acid, base, or specific enzyme to break the bond itself.
Why is water released when a peptide bond forms?
This is due to dehydration synthesis. The carboxyl group gives up an -OH (hydroxyl), and the amino group gives up an -H (hydrogen). Together, H + OH forms H2O (water), which is released as a byproduct when the carbon and nitrogen connect.
Are peptide bonds rigid or flexible?
The bond itself (C-N) is rigid and flat due to resonance. However, the bonds surrounding it (the alpha-carbon connections) are free to rotate. This combination of "stiff links" and "rotating joints" allows proteins to fold into complex 3D shapes.
How do researchers synthesize peptide bonds in a lab?
They use a process called Solid-Phase Peptide Synthesis (SPPS). They anchor the first amino acid to a resin bead and use chemical activators to force the peptide bonds to form one by one, washing away impurities between each step.
Conclusion
The peptide bond is the unsung hero of biology. It is the chemical handshake that allows individual amino acids to team up and perform complex functions, from building muscle to regulating blood sugar.
Understanding this peptide-peptide connection is the first step in mastering biochemistry. Whether you are studying the degradation of a research compound or the synthesis of a new drug, it all comes down to the strength and stability of that single, vital amide linkage.
Official Medical Disclaimer
The information provided in this guide is for informational and educational purposes only.
Peptides and GLP-1 research compounds are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. All research should be conducted by qualified professionals in a controlled environment. The statements regarding these products have not been evaluated by the Food and Drug Administration (FDA). Always consult your local laws and institutional guidelines regarding the use of peptides in research. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.