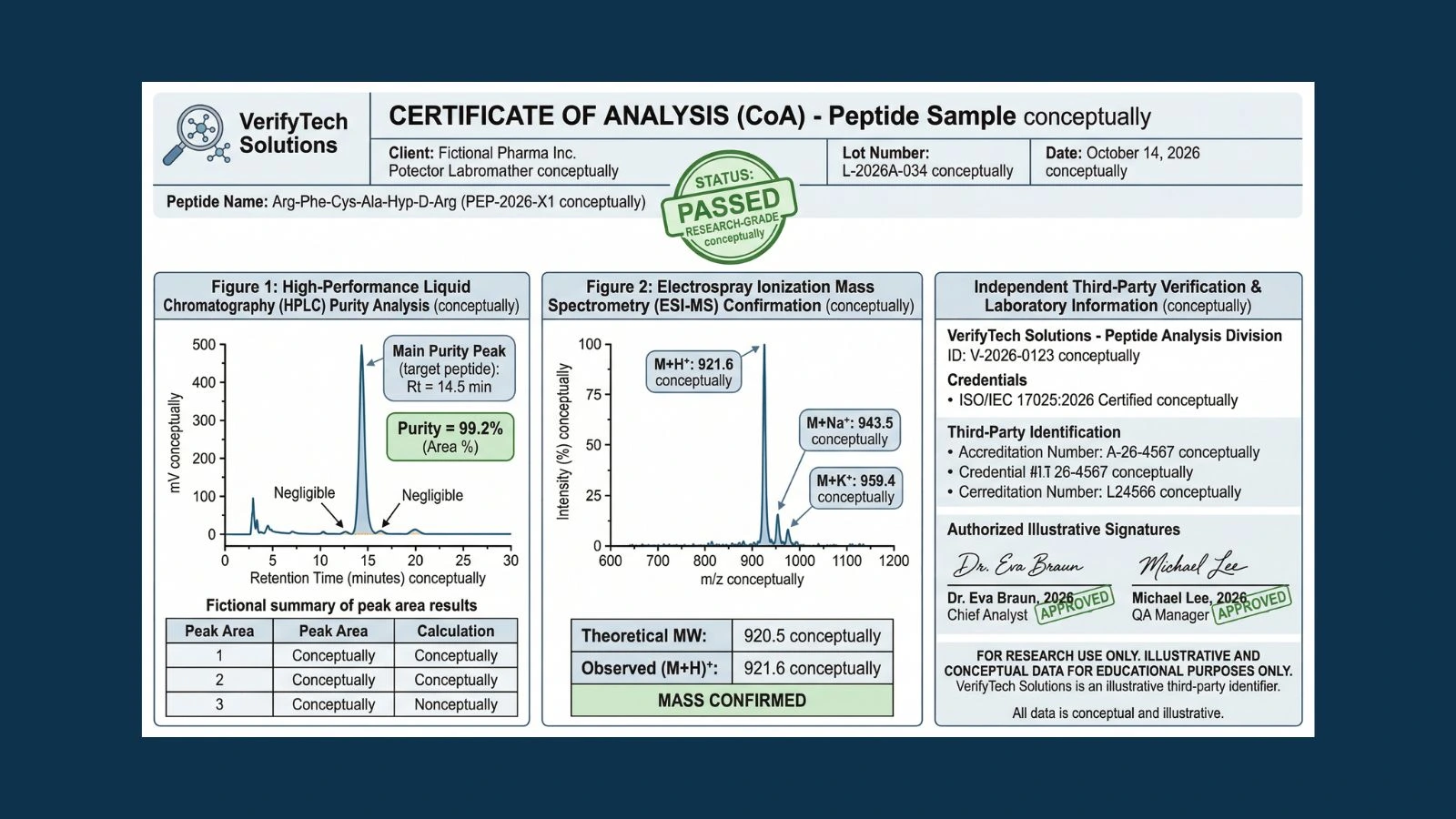
How to Verify Peptide Quality: Reading a Certificate of Analysis (COA)
Learn to read a peptide Certificate of Analysis: HPLC purity, mass spectrometry, endotoxin testing, and red flags that expose fake COA documents.
Understanding Peptide Certificates of Analysis: How to Read, Verify, and Trust the Data
A Certificate of Analysis is the single most important document in the research peptide supply chain. It is the only thing standing between a researcher and a vial of unknown contents. Yet the majority of people who buy peptides online have never actually read one carefully. They see the document exists, assume it is legitimate, and move on. This is a mistake that costs researchers reproducibility, time, money, and in some cases the validity of entire research programs.
The research peptide market has a real documentation problem. Vendors routinely provide what look like Certificates of Analysis that contain purity numbers, molecular weight data, and official-looking formatting, but which are either internally generated rather than third-party verified, reused across batches without lot-specific testing, or produced by laboratories with no accreditation and no analytical rigor. A COA that does not meet the minimum standards covered in this guide provides no meaningful quality assurance at all.
This guide explains what every section of a legitimate peptide COA means, how to distinguish real third-party documentation from vendor theater, what the analytical methods actually measure, what red flags to look for, and why each element matters to your research outcomes.
What Is a Peptide Certificate of Analysis?
A Certificate of Analysis is a formal document issued by a laboratory summarizing the analytical test results for a specific batch of a compound. It confirms that the batch was tested, describes the methods used, and reports the results against defined specifications. For research-grade peptides, the COA serves as the primary evidence that the compound in the vial matches the identity and purity stated on the label.
The key word in that definition is 'batch.' A legitimate COA is batch-specific, meaning it applies to one specific production run identified by a unique lot or batch number. It is not a generic company document, not a representative sample from a previous synthesis, and not shared across multiple products. The lot number on the COA must match the lot number printed on your product label. If those two numbers do not match, the COA tells you nothing about what is in your vial regardless of what the document says.
A 2025 review of analytical methods and regulatory guidelines for therapeutic peptide quality control, published via PMC (PMC11806371), describes how analytical characterization of peptides must confirm identity, purity, and stability, and that the ICH Q6B guidelines provide the international framework for what constitutes a complete quality characterization. For research-grade peptides, while full pharmaceutical compliance is not required, this framework defines the scientific minimum that meaningful quality assurance must address.
The Anatomy of a Legitimate Peptide COA: Section by Section
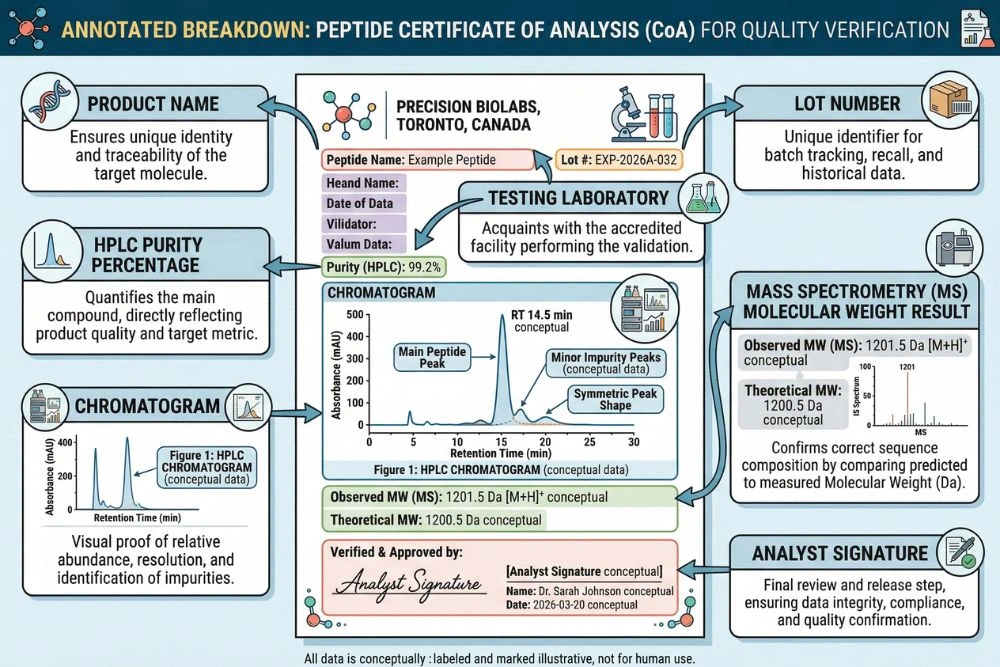
A complete peptide COA typically contains the following elements. Each one serves a specific purpose, and the absence of any of them is a meaningful signal about the document's quality.
1. Product Identity and Lot Number
The header of the COA should clearly state the peptide name or sequence, the product's catalog or batch reference number, and the lot or batch number that connects the document to a specific production run. The lot number on the COA must match the lot number on the product vial label. This is the chain-of-custody link that makes the document relevant to your specific product. Without this match, the COA is a generic document with no specific applicability to what you received.
Some COAs also include the peptide's molecular formula, molecular weight, and amino acid sequence. These are useful reference points but are not on their own quality indicators. They tell you what the vendor intended to produce, not what they actually produced. That is what the testing data confirms.
2. Testing Laboratory Identification
This is one of the most critical and most commonly overlooked elements of a COA. The document must clearly identify the laboratory that performed the testing, and that laboratory must be an independent third party with no commercial relationship to the vendor. Vendor self-testing has no scientific validity as an independent quality guarantee. A vendor who tests their own products and reports favorable results has an obvious financial incentive to do so.
The laboratory name, accreditation details, and contact information should appear on the document. Ideally the lab will be accredited to ISO/IEC 17025, the international standard for testing and calibration laboratory competence. This accreditation means the lab's methods, personnel, and equipment have been independently validated to meet a defined technical standard. When you see an ISO/IEC 17025 accredited lab on a COA, you have meaningful assurance that the testing methodology is sound.
The COA should also include a dated signature from an authorized analyst at the testing laboratory. This signature constitutes a professional attestation that the testing was performed and the results are as reported.
3. HPLC Purity Data
The purity percentage is the most commonly cited figure from a peptide COA, and it is generated by High Performance Liquid Chromatography, or HPLC. Understanding what this number actually measures is essential for interpreting it correctly.
HPLC works by forcing a dissolved sample through a column filled with a stationary phase, typically a C18 reverse-phase material, while pumping a gradient of solvents through at high pressure. Different compounds in the sample travel through the column at different speeds based on their chemical interactions with the stationary phase. As compounds exit the column, they pass through a UV detector, typically monitoring at 210 to 220 nanometers, where the detector records the absorbance of each compound as a peak on a chromatogram. Purity is then calculated as the area of the main peak divided by the total area of all peaks, expressed as a percentage.
A comprehensive analysis of peptide quality standards published via PMC (PMC10338602) describes this approach in detail, noting that RP-HPLC is used to determine lot homogeneity, stability, identity, content, and purity for peptide reference standards, with triplicate injections used to determine main peak content. The standard methodology monitors at 220 nm with well-defined column conditions that can be replicated independently.
The COA should not just report a purity number. It should include the actual chromatogram image, showing the full peak profile across the chromatographic run. A single dominant main peak with small or absent secondary peaks indicates high purity. A chromatogram showing multiple peaks of comparable size, poorly resolved peaks, or a broad elevated baseline indicates either a low-purity product or a synthesis that did not complete cleanly.
The analytical conditions used for the HPLC run should also be specified on or accessible from the COA: column type and dimensions, mobile phase composition, gradient program, flow rate, column temperature, and detection wavelength. Without this information, the purity figure cannot be independently reproduced or verified. A COA that shows only a number without the underlying analytical conditions provides no basis for scientific confidence in the result.
The research-grade threshold for peptide purity is generally considered to be 98% or above. Below this level, uncharacterized impurities begin to constitute a meaningful portion of the sample. Impurities in peptide synthesis most commonly include truncated sequences, deletion peptides, incompletely deprotected sequences, and oxidized residues. Any of these impurities are biologically active to varying degrees and can produce signals in cellular and animal assays that are independent of the peptide being studied.
4. Mass Spectrometry Identity Confirmation
Purity alone, even measured precisely by HPLC, does not confirm that the compound in the vial is the compound on the label. A pure sample of the wrong peptide could produce a single clean HPLC peak while being entirely the wrong compound. This is where mass spectrometry becomes indispensable.
Mass spectrometry determines the molecular identity of a compound by measuring the mass-to-charge ratio (m/z) of its ionized molecules. For a peptide, this produces a spectrum with one or more molecular ion peaks corresponding to different charge states of the same molecule. The observed molecular weight is then compared to the theoretical molecular weight calculated from the peptide's amino acid sequence.
Modern electrospray ionization mass spectrometry (ESI-MS) can determine peptide molecular weights with precision better than 0.1 Daltons on small peptides. This precision is sufficient to detect amino acid substitutions, deletions, modifications, and oxidation events that would change the molecular weight. A COA that shows an observed molecular weight matching the theoretical value to within instrument tolerance provides strong evidence that the primary compound has the correct sequence.
Recommendations published via PMC (PMC4830481) specify that purified peptides must include both MS data (preferably MS/MS data as well) and HPLC-UV chromatogram profiles to constitute adequate quality documentation. MS/MS fragmentation data provides sequence confirmation in addition to molecular weight matching, offering the highest level of identity assurance available from standard analytical methods.
The mass spectrometry data on a COA should include the theoretical molecular weight of the peptide, the observed molecular weight, and the difference between them. It should also specify the ionization method (ESI or MALDI) and ideally include a spectrum image or the raw m/z data. A COA that simply states 'Mass confirmed' without numerical data has provided no useful information.
The Endotoxin Problem: Why Purity and Identity Are Not Enough
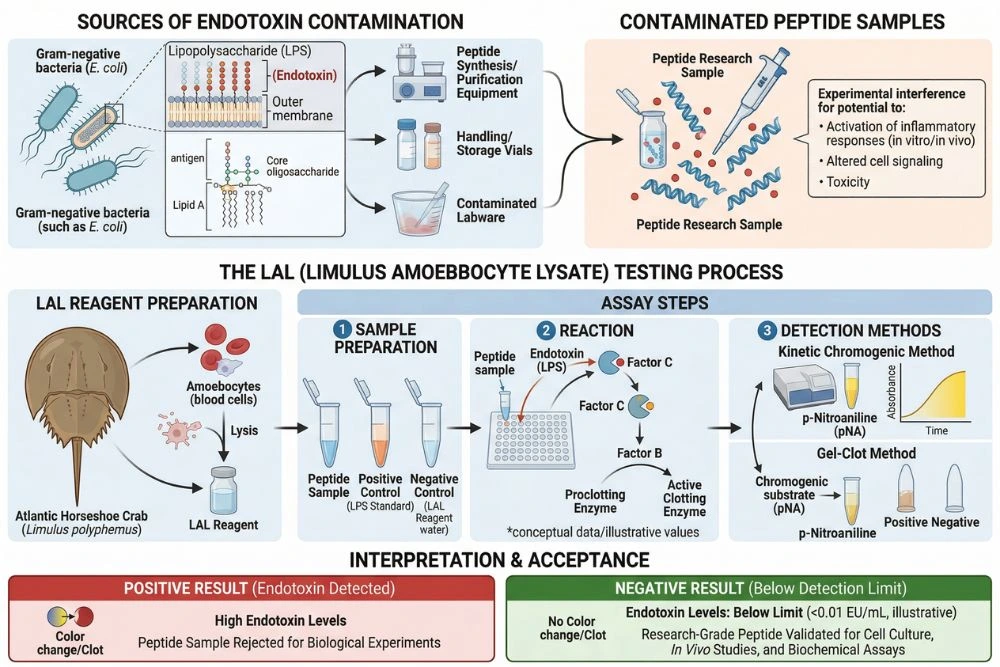
A peptide can pass HPLC purity testing at 99% and mass spectrometry identity confirmation and still contain a contaminant that invalidates every biological experiment it is used in. That contaminant is bacterial endotoxin.
Endotoxins, also called lipopolysaccharides or LPS, are components of the outer membrane of gram-negative bacteria. They are shed when bacteria reproduce or die and contaminate surfaces, equipment, and solutions that come into contact with bacterial material. What makes endotoxin particularly problematic is that it is completely invisible to HPLC and mass spectrometry. A COA showing 99% purity by HPLC tells you only about peptide content relative to other UV-absorbing species. It says nothing about endotoxin levels.
Endotoxin is among the most potent immune activators known. It activates TLR4 receptors on immune cells, triggering cascades of inflammatory cytokines. In cell cultures, nanogram quantities of endotoxin contamination produce inflammatory signals that dwarf most research compounds. In animal models, endotoxin contamination alters virtually every immune and inflammatory readout.
The standard test for endotoxin is the Limulus Amoebocyte Lysate (LAL) assay. The LAL test is mandated by the FDA, the European Pharmacopoeia, and the Japanese Pharmacopoeia for injectable pharmaceutical products, and it is the gold standard referenced by researchers. For research peptides used in cell culture or animal studies, endotoxin testing by LAL provides a critical layer of quality assurance that purity testing cannot supply.
A peptide COA that includes endotoxin testing results, expressed in endotoxin units per milligram (EU/mg) or endotoxin units per milliliter (EU/mL), is operating at a higher documentation standard. For researchers working with immune signaling, cytokine responses, inflammation models, or cell types with TLR4 expression, endotoxin testing is not optional.
Net Peptide Content: The Number Behind the Number
HPLC purity measures the percentage of the main peptide peak relative to all UV-absorbing species in the sample. What it does not measure is the non-UV-absorbing material present in the powder: water, counterion salts, and other components that contribute to total weight.
Net peptide content (NPC) is the actual percentage of the weighed sample that is peptide, accounting for water and counterion content. A peptide with 99% HPLC purity might have only 70 to 85% net peptide content. When you weigh out a milligram of that peptide to prepare a solution, you are actually getting only 0.70 to 0.85 milligrams of active compound.
This distinction matters for dose-response experiments, receptor binding studies, and any research context where precise peptide concentration affects outcomes. A COA that reports only HPLC purity without net peptide content is missing information researchers need. Top-tier vendors will include amino acid analysis (AAA) results, which determine net peptide content by quantifying amino acid composition after hydrolysis.
COA Red Flags: How to Spot Inadequate or Fraudulent Documentation
Not all COAs are legitimate, and not all inadequate COAs are fraudulent. Some vendors produce poor documentation out of ignorance. Both produce the same outcome: a false sense of quality assurance for a product that may not meet research standards.
- No third-party lab identified. Vendor self-certification is not legitimate documentation.
- Purity number without a chromatogram. Numbers without chromatograms cannot be verified.
- No analytical conditions specified. Professional reports always list column, mobile phase, and detection parameters.
- Mass spec section says only "Confirmed" or "Pass". This is meaningless without numerical data.
- Same COA used for multiple lot numbers. Every batch requires its own COA.
- No date or an old date. Testing must reflect the current batch.
- No analyst signature. An unsigned COA has no professional accountability.
- No endotoxin data for injection-grade claims. Endotoxin testing is required for anything labeled sterile or injection-grade.
Going Further: Independent Verification Tools
Finnrick Analytics
Finnrick Analytics maintains a database of independently tested peptide samples. Searching for a vendor allows you to see third-party results for specific products, providing unbiased verification. These are considered among the most reliable independent quality signals available.
Janoshik Testing
Janoshik is an accredited lab that offers testing services and maintains a public database. Vendors with multiple recent results there show a track record of genuine batch submissions for third-party testing.
Independent Lab Testing
Researchers can always submit a sample independently for HPLC and MS verification via contract testing organizations or university facilities. This represents the highest verification level, though cost and time may apply.
Putting It All Together: The COA as Part of a Sourcing Protocol
A strong COA is necessary but not sufficient for vendor trust. Documentation quality exists within a broader context of vendor reputation and operational practices.
The most reliable vendors are those consistently verified by independent sources. Vendors whose products repeatedly match claims when tested by others have built reputational credibility that no single COA provides.
The 2025 regulatory environment in the US reinforced this. Several vendors who provided apparently adequate documentation were later found noncompliant when audited. Documentation is a proxy for quality, not a guarantee. That proxy strengthens with third-party lab accreditation and independent community verification over time.
For a guide to the vendors recommended by PeptidesFinder, see the Best Peptide Vendors guide. For specific compounds, the Where to Buy BPC-157 guide apply this verification approach to actual purchasing decisions.
Frequently Asked Questions
What is a peptide Certificate of Analysis?
A Certificate of Analysis (COA) is a formal lab document reporting the analytical results for a specific peptide batch. It confirms identity via mass spectrometry, purity via HPLC, and may include endotoxin and net peptide content data. The COA must match the lot number on your vial to confirm the results apply to your specific material.
What is the difference between HPLC purity and net peptide content?
HPLC purity measures the proportion of UV-detectable peptide versus impurities. Net peptide content measures what fraction of total powder mass is actual peptide, accounting for water and salts. A peptide can have 99% HPLC purity but only 75% net peptide content. Both metrics matter for accurate dosing and interpretation.
Why does the COA testing lab matter?
The identity of the testing lab determines whether the COA is independent verification or self-reporting. A third-party, ISO/IEC 17025 accredited lab provides genuine independent assurance. A vendor’s own lab does not.
What HPLC purity level should I expect for research-grade peptides?
Research-grade purity is typically 98% or higher. Lower purity introduces meaningful impurity fractions that can affect research results. Pharmaceutical-grade standards are stricter. For preclinical research, 98%+ purity verified by an independent lab with MS confirmation is the baseline.
Can I trust a COA that shows 99% purity but no mass spec data?
Not fully. HPLC measures purity, not identity. A compound may be pure but incorrect. Mass spectrometry confirms the molecular identity, providing essential complementary verification. Without MS data, the COA is incomplete.
Does a high purity COA mean the peptide is endotoxin-free?
No. Endotoxins are not detected by HPLC. A peptide can have 99% HPLC purity and still contain biologically active endotoxins. Endotoxin testing via LAL assay is mandatory for peptides used in biological research.
What is the lot number and why does it need to match?
The lot or batch number identifies a specific production run. Each has slightly different purity and impurity profiles. The COA lot number must match the vial label for results to apply to your specific product.
Conclusion: A COA Is Only as Good as Its Weakest Element
A well-constructed peptide Certificate of Analysis tells a complete story: which lab tested the compound, which batch was tested, what methods and numerical data were used, and that an accredited analyst verified the results. Each element matters. Missing any weakens it from true quality documentation to marketing material.
Critical reading of COAs allows researchers to identify inadequate documentation, avoid unreliable vendors, and choose compounds whose quality is genuinely verified, not merely claimed.
For practical vendor evaluations, see the Peptide Vendors guide.
Don't forget to keep an eye out for our upcoming blog post: Sermorelin Benefits: How It Restores HGH Levels Naturally, covering clinical background and research findings for one of the most established growth hormone peptides.
Official Research Disclaimer
The information provided in this guide is for informational and educational purposes only. Research peptides are sold as research chemicals and are not FDA-approved for human use. They are intended strictly for laboratory research and are not for human consumption or for the diagnosis, treatment, or prevention of any disease. This article does not constitute medical advice. The author and publisher disclaim any liability for adverse effects resulting from the use or application of the information contained herein.